
Owing to the extreme complexity in vivo, the performance of elaborately designed anticancer formulation in the final clinical trials is often compromised from the previous experimental results. Such a inconsistency apparently reduces the druggability of the research objects and increases the risk of new drug development. Therefore, it is urgent to change traditional ideas of research and development for anticancer formulation.
In order to solve this problem, we have integrated the structure, function and program of biological systems into the design, and developed new preparation processes to construct a series of biomimetic anticancer formulations. For example, we developed in situ drug loading technology based on the unique hollow-porous structure of cage proteins. The high expression of corresponding receptors on the surface of tumor cells further enabled us to achieve targeted drug delivery. Meanwhile, a new hydrothermal process was developed to precisely regulate the bacterial structure for the accommodation of tumor antigen. Through natural infection, a large amount of antigen could be delivered to the dendritic cells, leading to a potent immune response. In addition, we also developed a new membrane emulsification process to coat the nanoparticles with cell membranes, thereby endowing them with excellent in vivo fate, such as long circulation and tumor penetration. Such a camouflage approach could significantly improve the performance for cancer imaging, diagnosis and chemotherapy.
As aforementioned, biomimetic formulation followed the intrinsic transport route in vivo, and precisely delivered drug, antigen or probe to the target site as expected. We believe these candidates will lead to the slightest adverse reaction, obtain the optimal application effects, reduce the risk of research and development, and promote the clinical conversion.
Controlling dispersion stability and assembled structures of nanoparticles in solvents during processing nanoparticle-based composite materials is one of the most important key factors toward designing the material properties. Although various surface designs have been reported up to date to achieve the improvement of nanoparticle dispersion stability, most system suffers from limited conditions to have enhanced dispersion stability (i.e. limitation in dispersible solvents and nanoparticle species). Herein, we report a new class of polymer dispersants which can be applied to various combinations of particles and solvent species. The proposed dispersant design involves the partial complex formation of polyethyleneimine (PEI) with functional anionic surfactant, which branches into polyethylene glycol based hydrophilic chain and alkyl based hydrophobic chain near the head group. The designed PEI-complex found to effectively adsorb on various species of particles including metal oxides, metal nitrides, metals, and carbon related materials. The particles fully covered with the designed dispersant were able to be dispersed into various series of solvents such as methanol, methyl ethyl ketone, ethyl acetate and toluene. Due to the high versatility of the designed dispersants, nanoparticles were able to be stabilized though relatively complicated process, such as ligand exchanging process of oleylamine stabilized Ag nanoparticles and simultaneous surface modifications of gas phase synthesized SiO2 nanoparticles during bead milling treatments. As well as the dispersion properties of PEI-complex-stabilized nanoparticles, some of our achievements toward aligning the surface modified nanoparticles on template particles/nanofibers and micro-structural control of composite materials will be introduced.
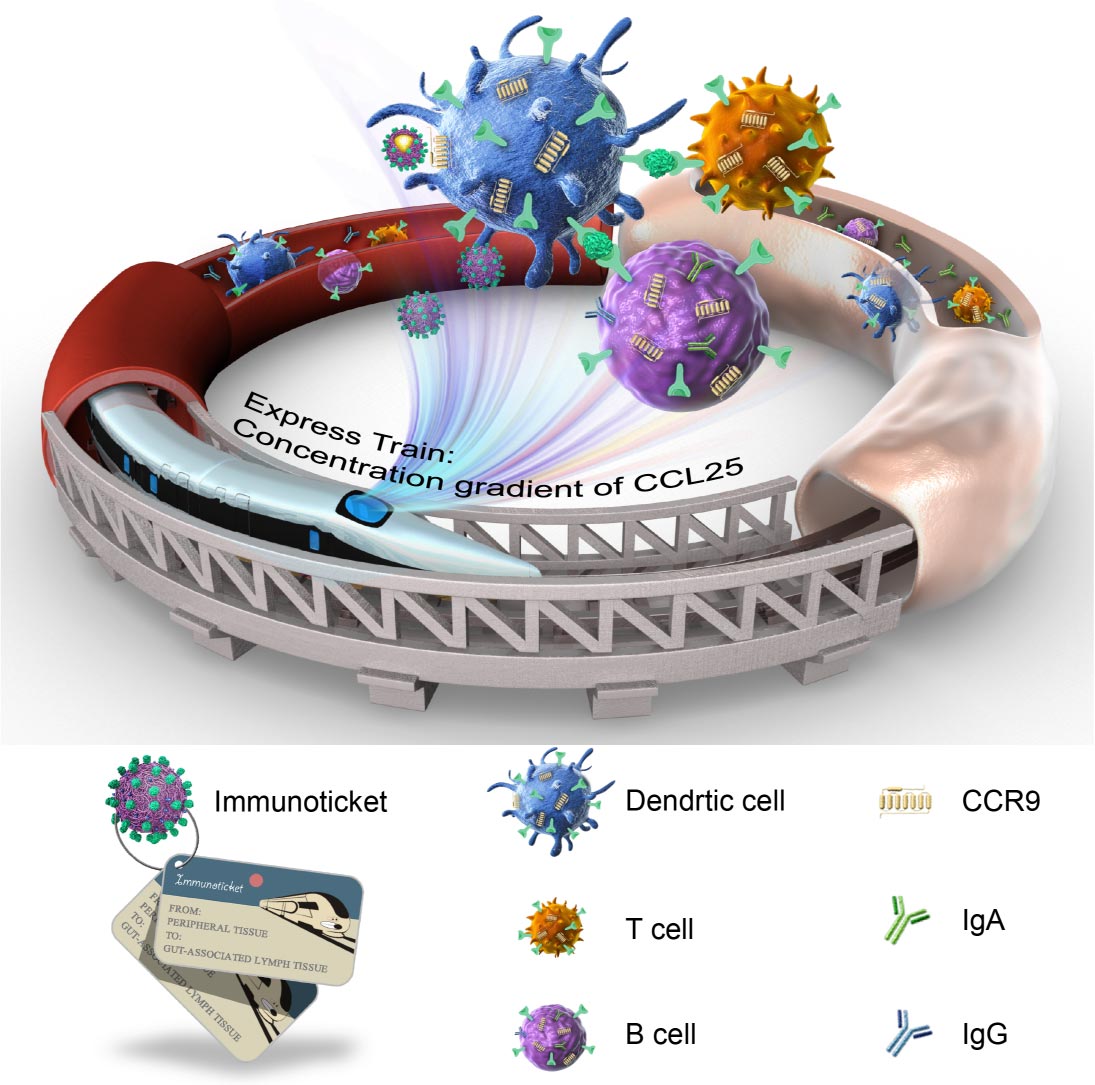
As peripheral lymphocytes are typically excluded from the gastrointestinal lymph tissues, current parenteral vaccinations failed to simultaneously induce systemic and mucosal responses. To break the natural barrier, we developed and heralded “immunoticket” capsules. Via internal phase separation, the capsules were formed with positive charged shells and oily core (oil-in-polymer particles) to spatiotemporally deliver antigens and all-trans retinoic acid (RA). After intramuscular vaccinations, these capsules functioned as immunoticket to cultivate the peripheral DCs with chemokine receptor 9 (CCR9). By hitchhiking on the concentration gradient of chemokine (C-C motif) ligand 25 (CCL25), the primed DCs would home to the gut associated lymphoid tissues (GALTs) and induced antigen-specific IgA secretion and T cell engagements. Compared with the currently employed RA-involved formulations, the immunoticket capsules stimulated enhanced RA-mediated gut-tropism by mounting the inflammatory innate immunity. Through controlling RA payloads, the potential regulatory T cell engagement was circumvented. In OVA and EV71 vaccinations, the immunoticket capsules induced potent serum IgG titer, antigen-specific cytotoxic T cells in the peripheral lymph tissues, as well as robust IgA secretion and T cell engagements on gastrointestinal sites. Our data suggested the potential of the immunotickets to serve as facile, effective and safe strategy to provide comprehensive immune responses against gastrointestinal infections and diseases.
Using delivery system shows promising signs on biobased therapy. However, there's a huge challenge for incorporating all the bioactive factors into one single carrier, leading to unsatisfied efficiency for present antigen/drug delivery system (A/DDS). To break this bottleneck, we uncover the cellular response that induced by graphene oxide (GO) and establish specific A/DDS based on this two dimensional material. On one hand, the flat micro GO exhibit unique properties, including extraordinarily high level of antigen adsorption, intracellular folding effect, and autophagy induction. Such a “One but All” vaccine delivery system induces high level of anti-tumor responses in a programmable way and finally results in the in vivo tumor regression. On the other hand, we evidence sandwiched graphene-cell membrane superstructures in different cells and reveal the transport of GO varies from Brownian to Lévy and even directional dynamics. In terms of above unique advantages, the applicability of sandwiched GOs in enhanced efficiency of membrane-specific drug delivery is thus demonstrated. Our findings inform approaches to program two-dimensional nanomaterials towards advantageous tumor vaccine delivery and intramembrane drug transport.
References:
[1] Pengyu Chen#, Hua Yue#, Xiaobo Zhai, Zihan Huang, Guanghui Ma, Wei Wei, and Li-Tang Yan. Transport of graphene nanosheet sandwiched inside cell membranes. Sci Adv, 2019. Accepted (# Authors contributed equally)
[2] Hua Yue, Wei Wei, Zonglin Gu, Dezhi Ni, Nana Luo, Zaixing Yang, Lin Zhao, Jose-Antonio Garate, Ruhong Zhou, Zhiguo Su, Guanghui Ma. Exploration of graphene oxide as an intelligent platform for cancer vaccines. Nanoscale, 2015, 7, 19949-19957 (Back cover story)
[3] Hua Yue, Wei Wei, Zhanguo Yue, Bin Wang, Nana Luo, Yongjun Gao, Ding Ma, Guanghui Ma, Zhiguo Su. The role of the lateral dimension of graphene oxide in the regulation of cellular responses. Biomaterials, 2012, 33(16): 4013–4020