
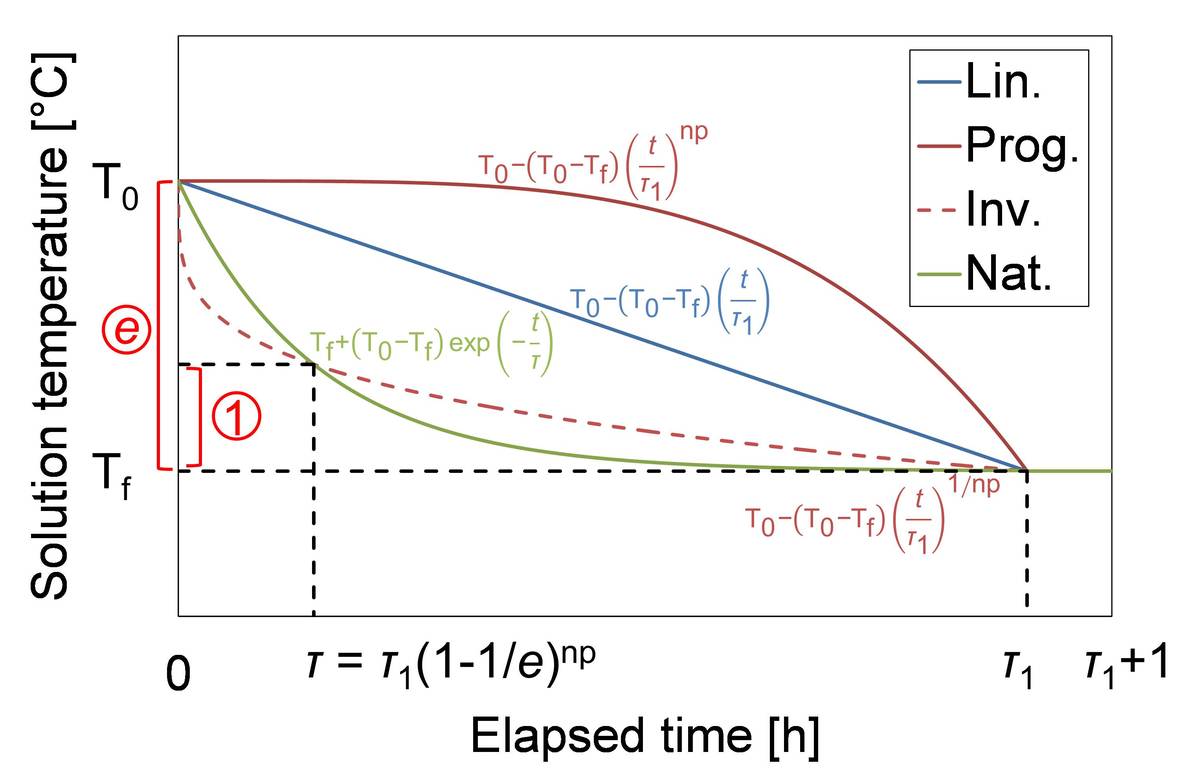
In the process of manufacturing crystal products, the crystal size distribution (CSD) of product crystals is required to be monodispersed and unimodal for improving productivity. Therefore, unintentional nucleation needs to be avoided in batch cooling crystallization because it leads to broad or bimodal CSD. Full seeding and partial seeding are well known as seeding policies for inhibiting or controlling nucleation. In full seeding, a large amount of seed crystals are added to inhibit secondary nucleation. On the other hand, in partial seeding, a small amount of seed crystals are added to trigger secondary nucleation and inhibit primary nucleation. In this study, partial seeding was mainly investigated. In the previous study, the optimum operating condition for partial seeding was successfully estimated based on coefficient of variation (CV) by utilizing computation results on inorganic model substance. Product CV was assumed to be small when seeding was effectively performed. Also in this study, the optimum operating condition and the resulting product properties were estimated for various cooling and seeding policies by utilizing computation results on inorganic model substance. Cooling policy was changed not only in the total batch time but also in the cooling mode, as seen in Figure. Here, linear cooling, programmed one, and natural one were performed. The order of programmed cooling np was set to 4 for small supersaturation in an early stage of crystallization. For improving symmetry of condition, the time constant of natural cooling was set to the same value as that of inverse programmed cooling the order of which was 1/np. Seeding policy was changed not only in the mean mass size but also in the CV. The population density of seed crystal was given by the convex quadratic function. Potassium sulfate was utilized as the inorganic model substance.
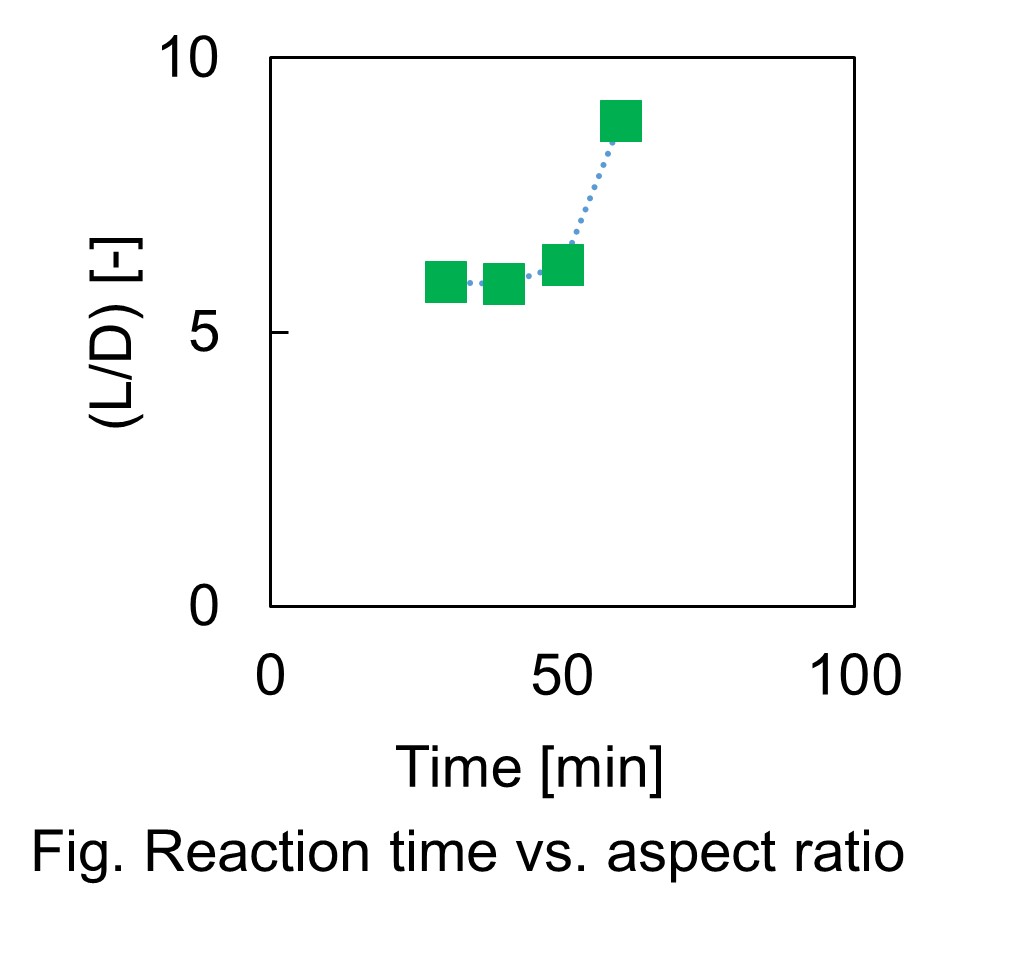
Transparent conductors are widely used for smart-phones, organic light emitting diodes and organic photovoltaics. Indium tin oxide (ITO) is the most common because of its high transmittance and conductivity. However, ITO have some defects. ITO is produced by a vapor phase synthesis and the rate is low, which makes ITO expensive. Second, indium is scarce. Finally, ITO is brittle. Therefore, the alternative transparent conductors have been studied eagerly. Among the studied transparent conductors, copper nanowires (Cu NWs) are promising. It is because copper is more inexpensive and abundant than indium. Moreover, Cu NWs are produced by a solution method, which is economical. Moreover, Cu NWs are flexible and this is advantageous for OLEDs and OPVs. Cu NWs are synthesized by a copper salt, a reducing agent and a capping agent, which determines the property, especially habit of crystals. The widely used capping agents for Cu NWs are amines. In this research, double-jet reactive reaction is applied for process of Cu NWs synthesis. In our previous study, it was difficult to synthesize Cu NWs of desired aspect ratio. On the other hand, double-jet process could control aspect ratio of Cu NWs by controlling reaction time, and to find out that concentration of reducing agent affects aspect ratio of Cu NWs.
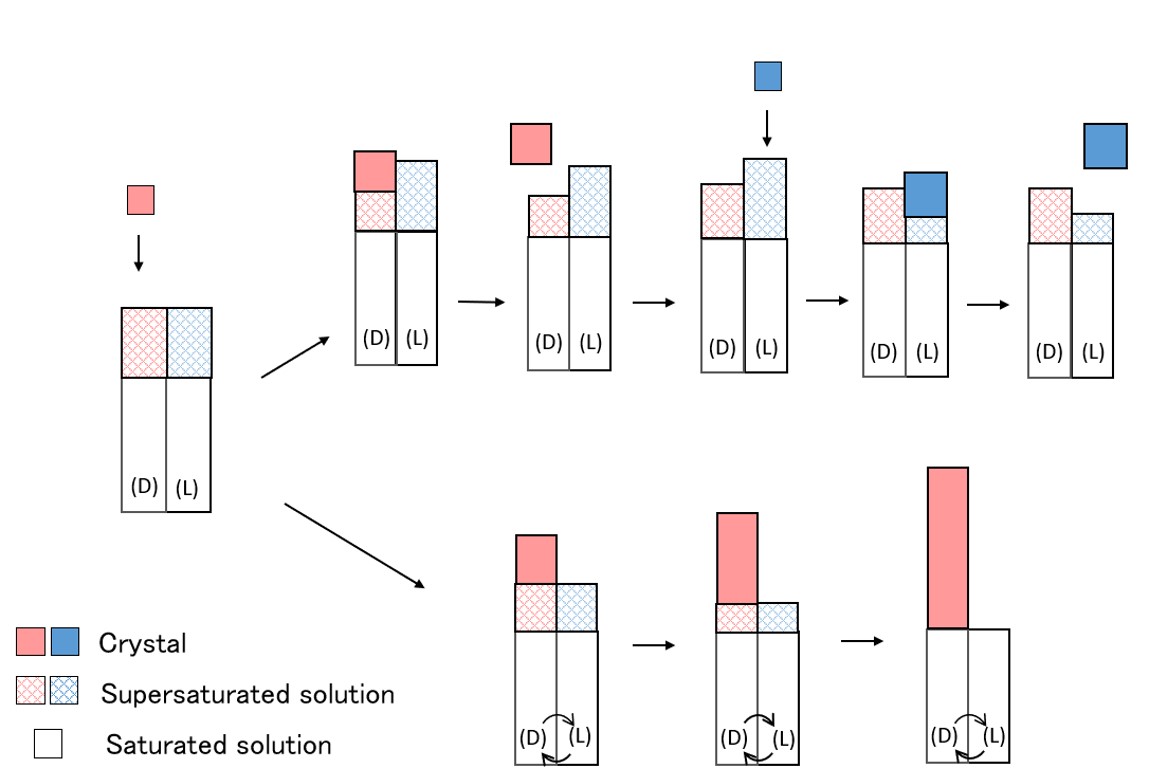
Many of the pharmaceutical compounds have asymmetric carbon. There is a need for techniques to reliably obtain one enantiomer. One of the methods of optical resolution is a preferential crystallization. In the preferential crystallization, crystals with high purity can be obtained by a simple operation. However, if chiral molecules do not racemize in solution, the maximum yield is 50%. By racemizing the chiral molecule in solution, other enantiomers can be converted to opposite enantiomer and crystallized as the desired product. So, the maximum yield would be 100%. Furthermore, repeated operations will be eliminated. In this study, the enzymatic reaction by racemase was examined as a method for racemization. We aimed to improve yield by performing preferential crystallization of DL-alanine combining it with racemization by alanine racemase. The alanine racemases gene from Streptococcus pneumoniae was artificially synthesized and expressed in E. coli shuffle T7. After several steps of purification, the enzyme of 35 kDa with electrophoretic homogeneity was obtained. The enzyme activity of alanine racemase was measured by monitoring the concentration of L-alanine with a circular dichroism dispersion meter. Specific activity for the conversion of L-alanine to D-alanine was 18.4 U/mg. Since alanine is crystallized as a racemic compound, it cannot be resolved by preferential crystallization. Thus, in order to crystallize as a racemic conglomerate, co-crystallization of alanine and p-chlorobenzene sulfonic acid (CBS) was performed. Optical resolution by preferential crystallization was performed. The alanine was dissolved at 40 °C and slowly cooled to 25 °C. When the solution reached to saturation temperature, seed crystals of D-Ala/CBS were added into the solution. After stirring for 1 hour at 25 °C, the crystals were recovered by filtration. The yield of crystals and the enantiomeric excess were 74% and 72%ee, respectively.
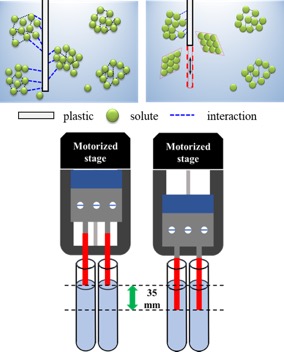
Since crystal nucleation directly affects the characteristics of the crystal, control of crystal nucleation is crucial in industrial crystallization. Previously, we reported that active diffusion of solute molecules in solution could be a trigger of nucleation. In this study, we investigated a new strategy to induce crystal nucleation. When immersing plastic pieces in solution, intermolecular interactions occur between the plastic surface and the solute molecules. When the plastic piece is moved vertically in this state, solute molecules which interact with the plastic also move together. The movement of the solute molecule reproduces unidirectional active diffusion of the solute molecule, and it is thought that crystal nucleation could be induced. In order to confirm this, we investigated the effect of the movement of plastic pieces on the crystal nucleation of L-alanine. Special experimental equipment and image processing software were developed. The plastic piece (polypropylene (PP)) was moved up and down vertically in alanine solution at a constant speed of 0.5, 2.0, and 5.0 mm/s. After that, the time when the first crystal was observed was recorded for at least 27 samples. Results were analyzed by cumulative distribution functions, and nucleation rate and average induction time were estimated. At all speeds, the nucleation rates were much higher than when the plastic was not in contact. In addition, it was also found that no crystals were generated when the plastic was immersed in the solution and left to stand. Therefore, it was suggested that the movement of plastic in solution induces nucleation of L-alanine. Comparing the nucleation rates at each speed, the nucleation rate was fastest at 2.0 mm/s. From these results, it was concluded that nucleation induction by the movement of plastic is caused through two processes, that is, the formation of intermolecular interaction and unidirectional movement of solute molecules.
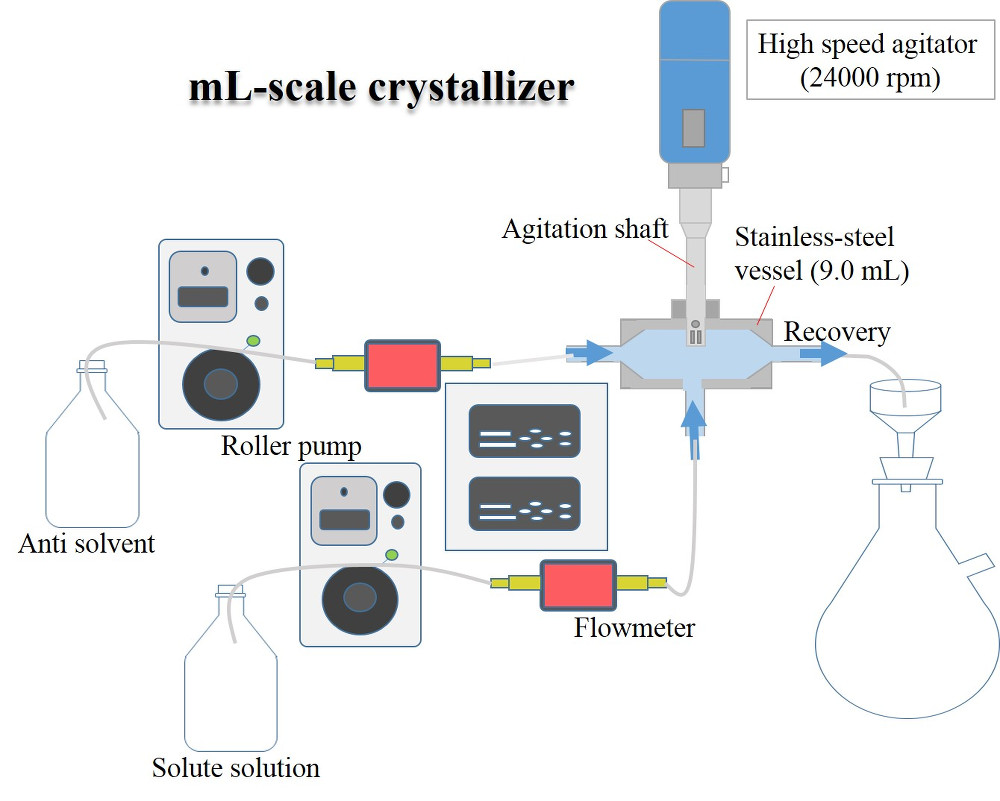
Novel small scale continuous crystallizer was developed to produce small uniform crystals. In the field of pharmaceutical crystallization, research on continuous crystallization is actively conducted. In particular, there is a need for a technique for continuously producing microcrystals without milling process. However, since the conventional continuous crystallizer has a large working volume, the residence time distribution becomes large, and it is difficult to control the crystal characteristics. Thus, we have developed a small continuous crystallizer to realize short residence time. The crystallizer is a 9 mL stainless steel device equipped with a high-speed agitator with a maximum stirring speed of 24000 rpm. Comparing with the large scale crystallizer, the average residence time is short, the distribution of residence times is narrow, and the concentration and temperature in the crystallizer are homogeneous. Therefore, small uniform crystals can be obtained. Anti-solvent crystallization of glycine was performed. Ethanol was used as a poor solvent. Almost 100% yield was attained even very short residence time (0.3-33 s). The shape and particle size of the obtained crystals were observed with a scanning electron microscope. More uniform crystals were observed compared to that obtained by the conventional crystallizer. Furthermore, it was also advantageous to polymorphic control. Due to the short residence time, the desired metastable crystals were recovered avoiding the solvent-mediated transition to the stable form.
Unseeded semi-batch cooling crystallization of potassium alum (AlK(SO4)12H2O, K-alum) was carried out to obtain uniform crystals with narrow size distribution. In this paper, influence of feeding condition on crystal size distribution is mainly discussed. Seeding policy studied by the research group of Iwate University is a powerful idea for the successful inhibition of secondary nucleation leading to monodisperse crystals. However, seeding methodology is often undesired particularly in the pharmaceutical and/or food industries because the possibility of containing impurity in the seed crystals is not always ignored. The internal seeding method (unseeded method) was reported by the above research group, however, the theoretical aspect of operation design has not been clarified. This study continues the internal seeding method to develop theoretical feed condition as unseeded crystallization policy. One of the strategy to obtain narrow size distribution is the division of cooling stage (feeding stage) for the separation of nucleation and growth period.
Polymorphism occurs when a compound can exist in more than one crystalline form. Because of the different properties of polymorphs, it is advantageous to choose the proper polymorph for the desired application. In the crystallization processes involving polymorphs, the formation of the desired polymorph has to be controlled and such a process should be robust and reproducible. In the polymorphic crystallization, relative nucleation rates, relative growth rates, and the transformation of crystallizing polymorphs should be controlled individually. Therefore, the determination of the crystallization (nucleation and growth) and dissolution kinetics, and thermodynamics are important for characterization of the crystallization behavior and transformation of the polymorphs. Thermodynamics determines the polymorphic forms and their properties such as solubility. The nucleation, growth and dissolution kinetics determine the kinetics of the crystallization and transformation of the polymorphs. In this work L-histidine (L-his) is the model substance; it is one of the essential amino acids and is significant in the food and feed industries. There are two known polymorphic forms, forms A and B. Form A is the stable form and form B is the metastable form. In the present study the crystal growth kinetics of the metastable polymorph B of L-his were performed and studied. The crystal growth rates were measured in aqueous solution of L-his. The crystal growth rates increased with increasing temperature, concentration of solute, and seed mass. The growth rates were correlated to the supersaturation with a power-low equation. The results indicated that the crystal growth process was controlled by surface integration mechanism at temperature of 10 and 25 degreeC and diffusion mechanism at 40 degreeC. Growth kinetics of the polymorphs of L-his will be used to begin characterization of the polymorphic transformations and the overall crystallization rate of L-histidine.
Crystallization is used widely in industrial processes for products such as chemicals and pharmaceuticals. Stirred-type crystallizers can produce various complex phenomena such as crystal growth, abrasion, and aggregation, which affect the total number of crystal particles in a vessel and their particle size distribution. Population balance analysis of the number of crystals coupled with mass balance of the solute material provides an important means of predicting and controlling the number of crystals in the crystallizer. In this study, particularly addressing the aggregation phenomenon, methods for measuring the degree of supersaturation in solution, the particle size distribution, and the amount of aggregation were developed. Relations among them were investigated for various seeding and cooling rate conditions.
Batch-type cooling crystallization of potash alum started with seeding was examined as a model case. The solution contains crystal particles in the vessel aspirated through the quartz observation cell every 10 min. A high-speed movie camera recorded the motion of crystals through the cell. The particle size distribution was calculated from the obtained image using the original MATLAB program. Moreover, the time evolution of solution concentration was estimated from measurements of the solution electrical conductivity and temperature. The solution conductivity is affected by the suspended crystal particles. Therefore, the effects of particle size and suspension concentration on conductivity were calibrated using unimodal glass particles.
Results show that the time evolution of the solution supersaturation degree and crystal aggregation were estimated successfully based on the combination of the solution conductivity, temperature, and particle size distribution measured during the crystallization operation. When the seed crystals were few, the crystal surface area was small during the initial stage of crystallization; then the mode diameter of crystals in the final stage became large.
Low aqueous solubility of crystalline drugs is the major issue in the pharmaceutical field. Normally, two-component amorphous solid dispersion is used as conventional strategy for improvement of their solubility. Unfortunately, it is known that amorphous phase shows poor stability. Therefore, development of novel strategy for improvement of solubility is strongly desired. On the other hand, our recent study using eutectic solid dispersion found that nucleation rate enhancement of second component encourages refining eutectic phase including target component. Normally, refining eutectic phase including target component is favorable structure for enhancement of dissolution rate. Therefore, it is suggested that enhancement of nucleation rate is probably effective strategy for improvement their solubility. In addition, it is expected that formation of eutectic having a refined microstructure occur with refining the eutectic phase, which affects their solubility. Therefore, the objective of this present study is development of newly strategy for improvement of dissolution rate by nucleation rate enhancement of second component based on eutectic melt system. Sorbitol (target component)-Xylitol (second component) was used in the experiment system. In this study, we focused on two nucleation factors that are shear force and supersaturation. Therefore, we investigated the influence of seed crystal mass and cooling rate on the nucleation of the second component and dissolution rate of target component. As a result, it is showed that dissolution rate of the crystal phase was influenced by the seed crystal mass and the cooling rate. Furthermore, it was found that the dissolution rate increased with increasing of seed crystal mass and cooling rate. This means that solubility could be improved by promoting the nucleation rate of the second component. Therefore, our novel concept based on nucleation rate enhancement of second component is probably effective strategy to improve the solubility of target component.
Crystallization operation is used as the final process in chemical products manufacturing. The production of crystals with high quality is required because the final product quality strongly depends on the crystal quality. Therefore, quality control in the crystallization operation is essential.
The crystal quality is determined by desired many kinds of characteristics. Generally, the average value of each characteristic of individual crystals is used as standard whether the desired quality is achieved or not. On the other hand, it is also important to evaluate the variation of their qualities constituting the crystalline particles. In particular, their qualities tend to vary when the agglomeration phenomenon occurs. Therefore, achievement of the desired quality and improvement of the variations in individual crystal quality is strongly needed to control in crystallization process. Our present work reported that homogeneity is a useful index to evaluate variations of crystalline particles. Thereby, the purpose of this research is to develop the newly method of crystallization operation for improvement of homogeneity for agglomerated crystalline particles in reaction crystallization. In this study, L-Aspartate sodium (L-AspNa) and hydrochloric acid (HCl) were used as reaction system. L-Aspartic acid is objective crystalline material.
As a result, we found that homogeneity could be improved by supersaturation modulation operation. Unfortunately, it was also showed that using only supersaturation as an operation variable is not enough to product crystals with various qualities because it directly influenced on their characteristics such as size and shape. For this reason, pH of the solution was used as additional operation variable for quality control of crystals because solubility of L-Asparatic acid depends on pH. Consequently, production of crystals having various qualities was succeeded with keeping high homogeneity. Therefore, it was suggested that crystalline particles with high homogeneity could be obtained by using the multiple operation variable.
Quality control in the crystallization operation is required to obtain desired products because its quality depends on the properties of crystalline particles. It is known that their properties such as morphology and shape are determined by nucleation and growth of crystal. Thereby, controlling the nucleation and growth process individually is important. Normally, nucleation and growth occur simultaneously in conventional crystallizers, which means that conventional crystallizer are not suitable to obtain uniform product. For this reason, development of novel crystallizer for controlling nucleation and growth individually is needed. On the other hand, designing a newly production process based on conventional crystallizer has some advantages because of low investment cost and standard operation. Therefore, it is suggested that a crystallization process specializing nucleation should be integrated to conventional process in serial to separate nucleation and growth. We focused on the Taylor Vortex. Previous study reported that Taylor Vortex shows high shear stress due to its specific vortex ring, which is favorable flow state for enhancement of secondary nucleation. However, the driving force of nucleation and growth is determined by a supersatutration of feed solution. This indicates that the balance of each processes is limited even if nucleation is separated from growth using two continuous crystallizers. On the contrary, we designed the novel cascade type crystallizer based on Taylor Vortex. The purpose of this study is investigation of operation strategy for designed cascade type crystallizer to produce crystalline particles with uniform quality. The crystal shapes obtained by conventional and novel cascade process were evaluated using SEM. As a result, the dispersion of crystal size became narrow, which means that quality of crystal could be improved by using our designed crystallizer. We expect that our proposed crystallizer is favorable continuous process to produce crystalline products with desired quality.
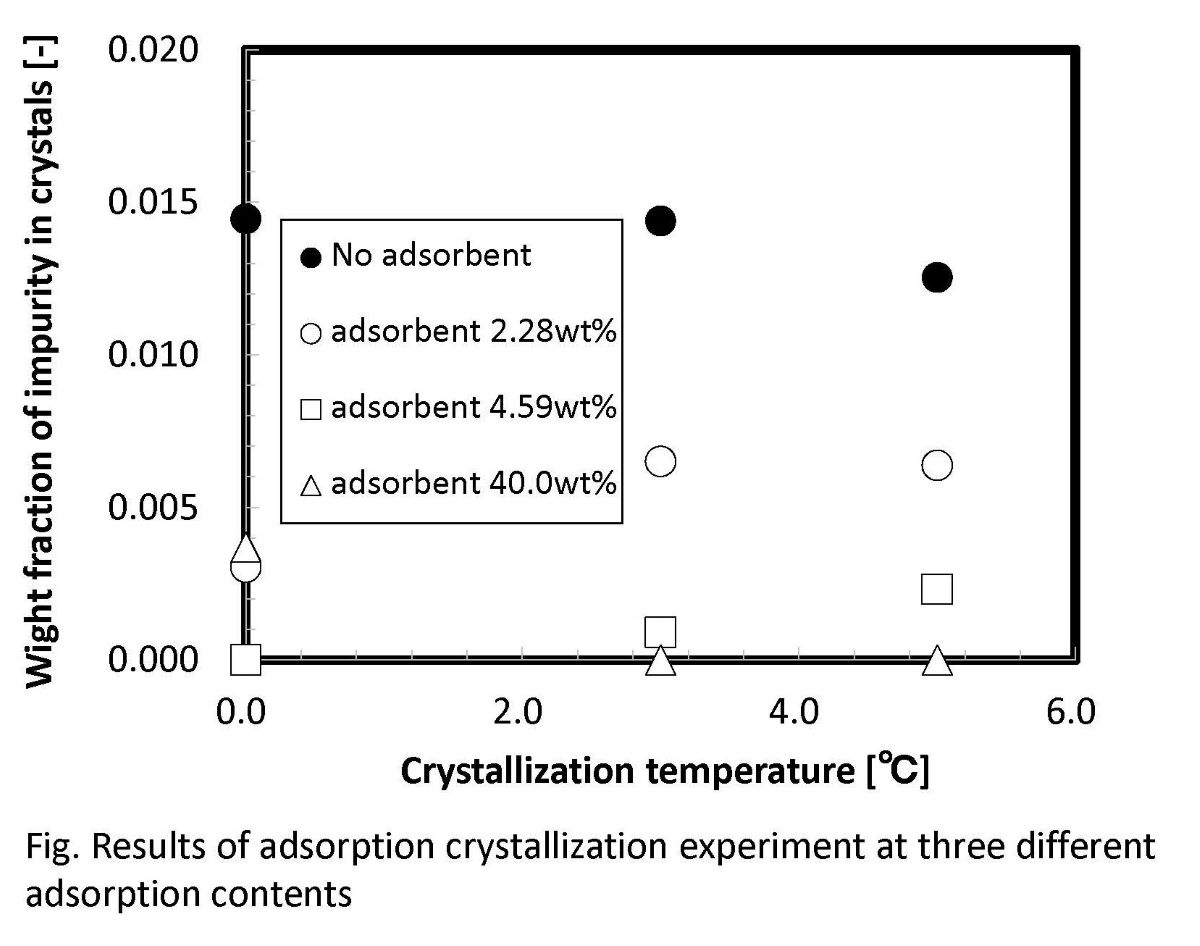
The adsorption experiments were first carried out to remove the impure acetic acid from the mixed acids (phosphoric acid + acetic acid) as raw model waste solutions. The adsorbent used in this study was very effective to adsorb acetic acid selectively. The adsorption capacity of adsorbent was about 5% for acetic acid. Moreover, new adsorption-assisted crystallization was performed for purification of phosphoric acid crystals from the mixed acids. The crystallization temperature and the additional ratio of the adsorbent was changed as the experimental conditions. It was found that crystals of purity more than 99% phosphoric acid were easily obtained. The yield of phosphoric acid crystals increased as the crystallization temperature was lowered. The purity of phosphoric acid crystals increased slightly when the additional ratio of adsorbent was increased in the mixed acids. It was clarified that high purity phosphoric acid crystals can be purified efficiently by the synergetic effect of the adsorbent.
Recovery of valuable resources from waste plating solution has been of great interest from the viewpoints of both environmental protection and resource management. In this study, we attempt reactive crystallization combined with ozone oxidation to recover phosphorus species, which are contained in a waste plating solution as the reducing agent for electroless nickel-phosphorus plating, as calcium phosphate. The effects of the experimental conditions such as pH, temperature, the amount of calcium chloride added to the solution on the recovery yield are examined.
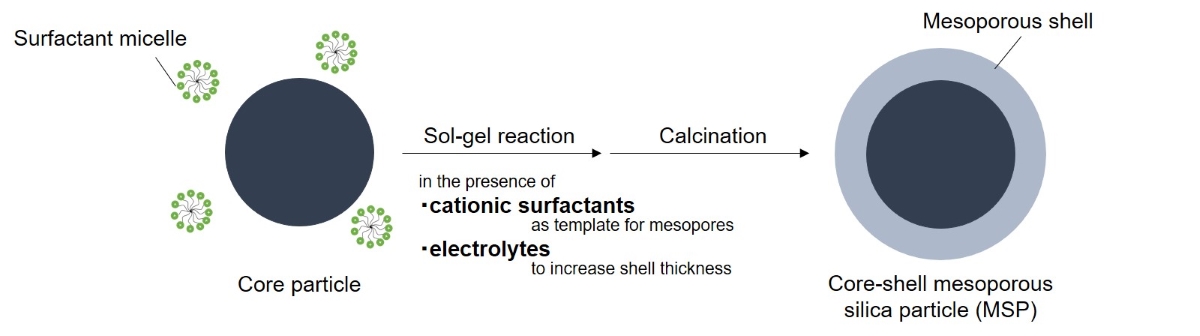
Core-shell mesoporous silica particles (MSPs) composed of a nonporous core and porous shell have attracted attentions due to higher monodispersity in particle sizes and more highly mechanical strength than mesoporous particles without a core. The core-shell MSPs can be used as packing materials to separate chemical substances in high performance liquid chromatography (HPLC) [1]. The performance of separation strongly depends on physical properties of MSPs, including particle sizes, pore sizes and shell thickness. The physical properties should be tuned to separate a variety of chemical substances.
Our group has been working on the synthesis of MSPs in sol-gel reaction where hydrolysis and condensation of silicon alkoxide are conducted in the presence of submicron-sized cores and cationic surfactants under basic conditions [2]. In conventional sol-gel reaction without cationic surfactants, sizes of silica particles formed are increased by electrolyte addition to the reaction system [3]. The increase in particle sizes by electrolyte addition has a potential of thickening mesoporous shell on the cores in the presence of cationic surfactants.
In this study, synthesis of core-shell MSPs was performed in the presence of electrolytes to increase the mesoporous shell thickness.
References
[1] Hayes R., Ahmed A., Edge T. and Zhang H., “Core–shell particles: Preparation, fundamentals and applications in high performance liquid chromatography”, J. Chromatogr. A, 1357, 36–52 (2014)
[2] Ishii H., Nara M., Hashimoto Y., Kanno A., Ishikawa S., Nagao D. and Konno M., “Uniform formation of mesoporous silica shell on micron-sized cores in the presence of hydrocarbon used as a swelling agent”, J. Sol-Gel Sci. Tech., 85, 539-545 (2018)
[3] Nagao D., Kon Y., Satoh T. and Konno M. “Electrostatic interactions in Formation of Particles from Tetraethyl Orthosilicate”, J. Chem. Eng. Japan. 33, 468-473 (2000)
Mesoporous silica are functional materials with high specific surface area caused by regularly ordered mesopores. Mesoporous silica have been applied to absorbents, catalyst supports and drug delivery carrier. In order to apply the mesoporous silica to various fields, particulate mesoporous silica is attracting a lot of attention. One of the important factor to be controlled is pore size distributions in the synthesis of mesoporous silica particles (MSPs). Micelles of surfactants can be a template for mesopores of silica particles formed in the sol-gel reaction [1, 2]. Addition of hydrophobic molecules as a swelling agent to the synthetic system could enlarge pores in the silica matrix [1].
Our group has been working on the synthesis of MSPs in sol-gel reactions in mixed solvents of water and ethanol in the presence of cationic surfactant used as template micelles [3]. The addition of swelling agents increased sizes of mesopores in MSPs [4], although mechanism on enlargement of mesopores in the formation of MSPs is not sufficiently clarified.
The present study examined the effects of solvent composition on spherical particle formation and pore size distribution. The affinity of swelling agents to the mixed solvent was examined using different hydrophobic molecules. Analysis by Hansen solubility parameter (HSP) could correlate swelling agents with pore size distribution.
[1] J. S. Beck et al., J. Am. Chem. Soc., 114, 10834–10843 (1992), [2] T. Yanagisawa et al., Bull. Chem.Soc.Jpn, 63, 988–992 (1990), [3] H. Ishii et al., Adv. Powder Technol., 27, 448–453 (2016), [4] H. Ishii et al., J. Sol-Gel Sci. Technol., 85, 539–545 (2018)
Characteristics of particles strongly depend on the particle shape. Synthesis of anisotropic particles has been extensively studied to functionalize particles in recent years. Location and orientation of the anisotropic particles can diversify colloidal assemblies of the anisotropic particles.
To fabricate the colloidal assemblies, several methods such as suspension drying and using chemical interaction between particles have been developed. Depletion interaction is one of the powerful approaches to assemble colloids into desired structures in solution. The attractive depletion interaction between particles is induced by osmotic pressure in the presence of depletants (e.g. non-absorbing polymer, surfactants and nanoparticles). The attractive interaction is proportional to both depletant concentration and overlapping between depletant layers to which depletants cannot access.
Our group previously reported depletion-interaction-driven assembly of golf ball-like particles for development of colloidal macromolecules [1]. Golf ball-like particles have a large number of dimples on their surface.
In the present work, we tried to synthesize golf ball-like particles having hemi-rod-shaped dimples and demonstrated an assembling of the golf ball-like particles and rod-shaped particles via depletion interaction. Anisotropic particles with rod-shapes are expected to be captured by the hemi-rod-shaped dimples due to depletion interaction. The capture of particles has a potential of new purification process in which targeting particles can be intentionally collected depending on their shapes and sizes.
[1] K. Watanabe, Y. Tajima, T. Shimura, H. Ishii, D. Nagao, Journal of Colloid and Interface Science, 534, 81-87 (2019).
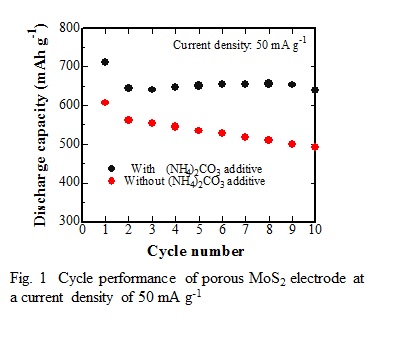
Recently, MoS2 is considered to be the most prospective candidate of anode materials for lithium ion batteries becuase of its high theoretical capacity (670 mAh g-1) and layered structure. However, it has usually suffered from structural deterioration, due to a large volume change on charge/discharge and low intrinsic electrical conductivity, which lead to a poor cycling performance. In this study, we have prepared porous MoS2 microspheres by spray pyrolysis with (NH4)2CO3 additive and investigated their pysical and electrochemical properties.
The precursor solutions were prepared by dissolving the required amount of (NH4)2MoS4 and (NH4)2CO3 in distilled water. Spray pyrolysis synthesis was conducted at different synthesis temperatures and concentrations of precursor solutions at a gas (3%H2+97%Ar) flow rate of 2 L min-1. The resulting samples were characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), and X-ray photoelectron spectroscopy (XPS). N2 adsorption–desorption measurements were performed at a liquid N2 temperature of 77K. Pore volume and size distribution were estimated from the desorption branches of the isotherms by the Barrett–Joyner–Halenda model. Their electrochemical properties have been investigated using CR2032 coin-type cells.
The XRD peaks of the sample prepared at 800°C from the precursor solution of 0.05 mol L-1 (NH4)2MoS4 and 0.50 mol L-1 (NH4)2CO3 could be indexed well to the hexagonal phase of MoS2 (ICSD No. 31067) without any other phases. The desired Mo-related bond features of the sample were confirmed by XPS analysis. The pore structure analysis based on N2 adsorption–desorption isotherm led that the MoS2 microspheres with pore size less than 200 nm could be prepared and the (NH4)2CO3 additive in precursor solution could enlarge the porosity in MoS2 microspheres, especially the pore size between 20 to 100 nm. As a result, the porous MoS2 microspheres improved its cycle performance, as shown in Fig. 1.
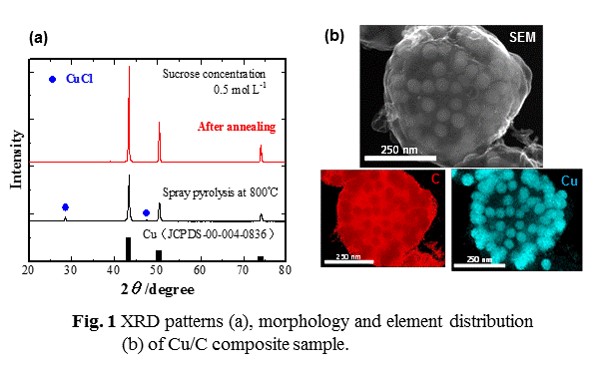
Porous carbon microspheres impregnated with transition metal-based nanoparticles have been considered as next generation materials for energy and environmental applications. In this study, we have prepared cupper (Cu) nanoparticles impregnated porous carbon microspheres by spray pyrolysis with heat treatment from the precursor solutions which appropriate amount of sucrose (C12H22O11) and cupper chloride dihydrate( CuCl2 2H2O) with citric acid (C6H8O7) additive were dissolved in distilled water, and investigated their pysical properties.
The precursor solutions were prepared by dissolving the required amount of C12H22O11,CuCl2 2H2O and C6H8O7 in distilled water. Spray pyrolysis synthesis was conducted for different synthesis temperatures and concentrations of precursor solutions at a gas (N2) flow rate of 2 L min-1. The as-prepared samples were further annealed at 800 °C for 2 h in a 3%H2/97%N2 mixture gas. The resulting samples were characterized by X-ray diffraction (XRD), field emission-scanning electron microscopy (FE-SEM) with an energy dispersive spectroscopy (EDS). Nitrogen (N2) adsorption–desorption measurements were performed at a liquid N2 temperature of 77 K The carbon content of the samples was estimated using an element analyzer (CHNS, Elementar, Vario Micro Cube).
Fig. 1a shows the XRD patterns of the sample prepared at 800 °C from the precursor solution by spray pyrolysis and then annealed at 800 °C for 2 h in a 3%H2/97%N2 mixture gas. It can be clearly seen from the XRD pattern of as-annealed sample that crystallized Cu could be obtained from the present preparation route. The N2 adsorption-disorption isotherm of the as-annealed sample indicated that it had mesopore structure, and the content of carbon was 28 wt% by CHNS analysis. Furthermore, it could be verified from the SEM-EDS analysis that the porous carbon microspheres impregnated with cupper nanoparticles could be sythesised by the present preparation route (Fig. 1b)
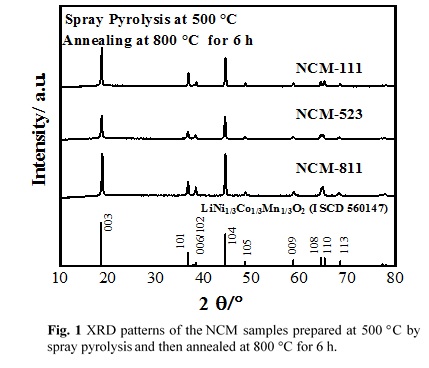
LiNixCoyMnzO2 (x≥0.33, x+y+z=1) (NCM) have a great potential as cathode materials for Lithium-ion batteries due to their abundant sources, high specific capacities and outstanding thermal stability. With the increasing content of nickel, NCM shows higher reversible capacity together with lower cost and toxicity. In this study, spherical nanostructured NCM particles were synthesized directly from homogeneous precursor solutions by spray pyrolysis followed by annealing in air atmosphere. The effect of process parameters on the physical and electrochemical properties of NCM was investigated.
LiNO3, Ni(NO3)2·6H2O, Mn(NO3)3·6H2O and Co(NO3)2·6H2O were used as the starting materials. These materials were dissolved in distilled water with the stoichiometric ratios of NMC. The starting solutions were atomized by an ultrasonic nebulizer at a frequency of 1.7 MHz. Then the generated droplets were carried to a spray pyrolysis reactor heated at different temperatures (400 -600 °C ) by the air gas with a flow rate of 2 L min-1. The as-prepared samples were further annealed at different temperatures in air atmosphere.
Crystal structures of the synthesized materials were determined by X-ray diffraction (XRD) analysis using Cu-Kα radiation. Fig. 1 shows the XRD patterns of the LiNi1/3Co1/3Mn1/3O2 (NCM-111), LiNi0.5Co0.2Mn0.3O2 (NCM-523) and LiNi0.8Co0.1Mn0.1O2 (NCM-811) samples synthesized at 500 °C by spray pyrolysis followed by annealing at 800 °C for 6 h. It can be seen that the well-crystallized NCM samples are formed with α-NaFeO2 hexagonal structure.
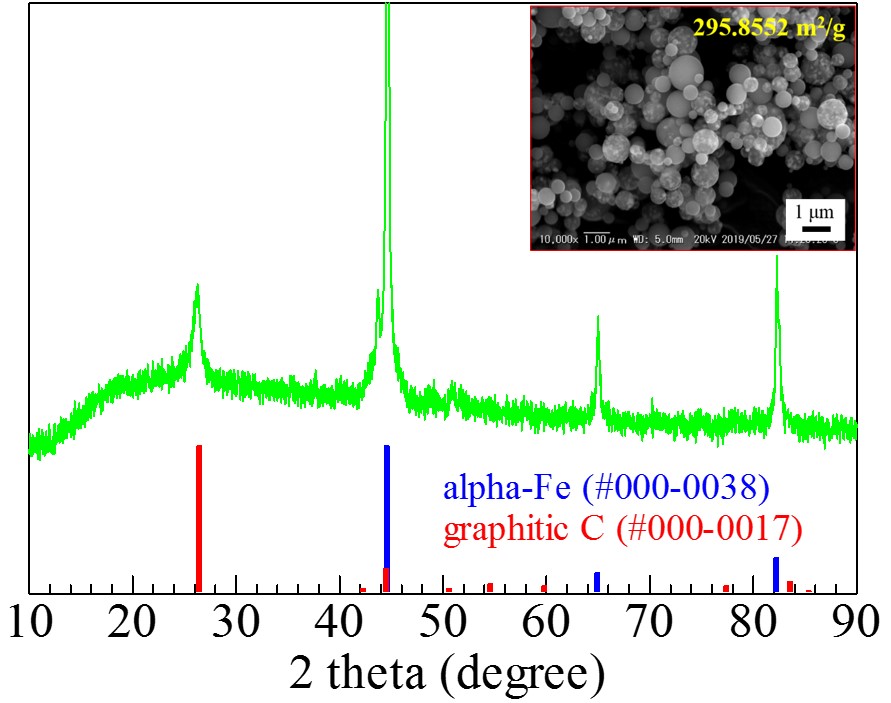
Integration of renewable energy sources with energy storage systems appears to be an encouraging approach to reach sustainability in green energy supply. It is expected that iron particles dispersed carbon microspheres (Fe@C) will find a broad application in many future generation energy storage systems and environmental issues. In this work, we have synthesized Fe@C using ultrasonic spray pyrolysis (USP) followed by annealing and studied synthesis parameters.
The precursor solution was prepared by dissolving an appropriate amount of sucrose, ammonium nitrate and iron nitrate in distilled water. Spray pyrolysis synthesis of Fe@C was carried out at 700 °C in an inert atmosphere (pure N2). Post-annealing of the samples was conducted at 700 °C for 4 hours in a reducing atmosphere (3%H2/97%N2). The obtained samples were characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), Fourier transform infrared (FTIR) spectra, and Brunauer-Emmett-Teller (BET).
Fig.1 shows the XRD pattern of the Fe@C prepared at 700 °C spray pyrolysis temperature followed by annealing at 700 °C for 4 hours. Three sharp peaks at approximately 45 °, 65 ° and 82 ° correspond to zero valent Fe while a peak at about 27 ° indicates the formation of graphitic carbon. SEM observation of the obtained sample shows that zero valent Fe particles evenly dispersed in the carbon microspheres (Fig.1 inset).
Gallium oxide (Ga2O3) is known to have five polymorphs, that are, α-, β-, γ-, ホエ-, and ε- Ga2O3. Among them, β-Ga2O3 is the most stable crystal structure and attracted recent attention as the material for power electronics, solar cells, and sensors. The conventional synthetic method of β-Ga2O3 nanoparticles is the so-called two-step method where hydrothermally synthesized GaOOH is calcinated at temperatures around 700 °C or above. In this presentation, we report an alternative one-step method to synthesize of β-Ga2O3 nanoparticles under solvothermal condition. In this method, gallium nitrate was dissolved in alcohol and then heated at 300 °C or above in a high-pressure reactor. The solid products were purified and analyzed by an X-ray diffractometer and a scanning electron microscope. Results showed that the crystal structure of the products gradually changed from γ-Ga2O3 to β-Ga2O3 as the reaction time increased. The average size of the produced nanoparticles was about 50 to 100 nm, which is much smaller than the products from the two-step method. This might be because our one-step method does not require a calcination process that allows the aggregation of particles during annealing.
Metal nanoparticles play an important role in catalysis, sensing, and imaging applications. Bimetallic systems are attractive for improving the physical and chemical properties by varying the elemental combination, composition ratio, and architectures of nanoparticles. However, designing the bimetallic nanoparticles are difficult because of many factors influences the nucleation and growth processes. In this study, we focused on the reduction rate of the metal precursors during the polyol synthesis of Ag-Rh nanoparticles. A microflow reactor was employed for obtaining reliable kinetic information by enhanced heat transfer performance. Reaction at elevated temperature in ethylene glycol was successfully examined by rapid heating and cooling with a microflow reactor. Ag precursor and Rh precursor showed strong interaction on the reduction rate. Reduction of Ag+ in ethylene glycol got slower with the higher reaction temperature, which contradicts to the Arrhenius law. The concentration profile of rhodium acetate would cause complex behavior due to its catalytic effect on the reduction. Elemental mapping of synthesized particles implied that the solid-state diffusion played a crucial role in the formation of alloyed Ag-Rh nanostructures.
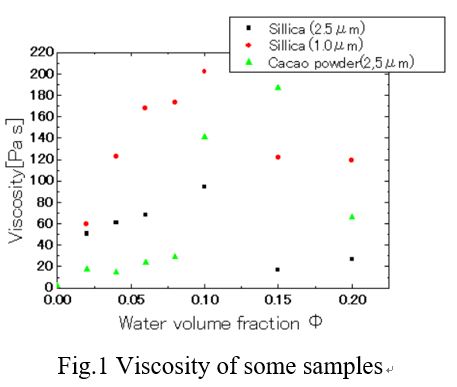
When a small amount of water is added to a suspension in which particles are dispersed in oil, the number water between particles is formed. Such a substance is called a capillary suspension, it having fluidity change to flow characteristics like gel. However, detailed mechanisms and characteristics of capillary suspension have not been studied. In this study, the particle / oil / water capillary suspension was prepared using various particles and evaluated the rheological properties.
Firstly the particles and the oil were mixed with a dissolver stirrer to prepare a suspension. Next, capillary suspension was prepared by adding small amount of water to the suspension and stirring for 10min. Viscosity and rheology of the product was measured with a rotary rheometer. Fig.1 shows the effect of water volume fraction on the viscosity of capillary suspensions prepared by using three types of particles (Sillica(2.5μm),sillica(1.0μm),Cacao powder(2.5μm). The viscosity profile shows the maximum value at the certein water volume fraction, which we called the optimum crosslinked water content (OCWC) . From the results, the samples using particles of the same material showed the same OCWC. However, samples using different particle with same particle size showed different OCWC. When the three-phase contact angle of cocoa powder is lower than that of sillica. It is considered that the OCWC became high because more hydrophilic characteritics. It is thought that the rheological property value of the capillary suspension can be estimated by the three-phase contact angle, interfacial tension, particle size and particle concentration. By measuring the yield stress, it is possible to calculate the capillary force acting between particles of each sample. It was found that the three-phase contact angle, which varies depending on the material of the particles, is closely related to the rheological property.
We focused on the interface between the polypropylene (PP) resin and the polyamide 6(PA6) resin, which was generated when the PA6 resin was located at the center of the carbon fiber reinforced polypropylene (CFRPP) to enhance the strengthen of the CFRPP. By adsorbing the polymer particles on the surface of the carbon fiber through the electrodeposition, the particles played a important roles to improve the interfacial adhesion between the PP and the PA6 resins. And also, the interface between CFRPP and metal was investigated to incorporate CFRPP to multi-material structures. polymer particles with the same component as the resin were adsorbed on the metal surface, and the particles were impregnated to fine defects on the metal surface, thereby improving the interfacial adhesion between CFRPP and the metal. Therefore, we examined the application of polymer particles to fabricate CFRPP for practical use.
A microlens array is the optical element which has many lenses with a diameter of several to hundreds micrometers on its surface. At present, the microlens arrays are prepared by the top-down processes such as photolithography and etching, where the productivity of the arrays is not very high. Moreover, it is difficult to obtain the arrays with smaller lenses through the top-down processes. To prepare the smaller microlens arrays efficiently, the bottom-up processes such as self-assembly are required. In the present study, we have tried to prepare the mesolens array with a diameter of hundreds nanometers from the non-close-packed (NCP) colloidal monolayers, which are prepared by the convective self-assembly (CSA) using a glass substrate and the silica particles grafted with cationic polyelectrolyte, poly(vinylbenzyl trimethylammonium chloride) (PVBTA). When the NCP colloidal monolayers are annealed, PVBTA would be removed and a part of the silica particles are thought to be melted. It is therefore expected that the silica particles and the glass substrate could fuse together to form the mesolens array, because they are mainly composed of silicon dioxide.
PVBTA were grafted by free radical polymerization on the surface of the monodisperse silica particles, and then the PVBTA-grafted silica particles and the glass substrate were applied to the CSA process to prepare the NCP monolayers. The obtained monolayers were annealed at 973 K in a muffle furnace for several hours. The annealed particle monolayers were observed by the atomic force microscope (AFM) and the scanning electron microscope (SEM). As a result, the height of the silica particles were found to decrease with an increase in annealing time, which probably indicates that partial fusion of the silica particle and the grass substrate proceeded as the annealing time increases.
Polymeric microcapsules are micron-sized particles with a liquid core and a polymer shell. The shell of the microcapsules can isolate the encapsulated substance from the external environment. Microcapsules exhibit various functions by combining different core substances with shell materials. In particular, microcapsules containing a phase change material have advantages such as an increase in heat exchange efficiency due to an increase in specific surface area and prevention of the encapsulated phase change material from leakage, which can be served as efficient heat storage materials for energy saving applications. Most of the microcapsules used as heat storage materials are made of melamine-formaldehyde materials (MFM). MFM have excellent mechanical strength and heat resistance. However, it has been reported that the resultant microcapsules contain unreacted formaldehyde which has high carcinogenic to human. There is therefore an increasing demand for reducing or removing formaldehyde from the products. In this study, we developed a process to prepare formaldehyde-free melamine microcapsules by using melamine, urea, glyoxal and 2,2-dimethoxyacetaldehyde (DME) [1] as the shell materials, based on the previous study [1] and optimized the preparation conditions for controlling the shell thickness. Specifically, we prepared a prepolymer solution and mixed the solution to oil-in-water (O/W) emulsion composed of Isopar G and aqueous solution containing poly(E-MA), as the oil and water phases, respectively [2]. We discuss the effect of glyoxal/DME molar ratio and NH2/CHO molar ratio on the formation of the microcapsules as well as the thickness of the microcapsules.
[1] G. Leon et al., RSC Adv., 7, 18962-18975 (2017)
[2] Eiji Kamio et al., Langmuir., 24, 23, 12387-13298(2008)
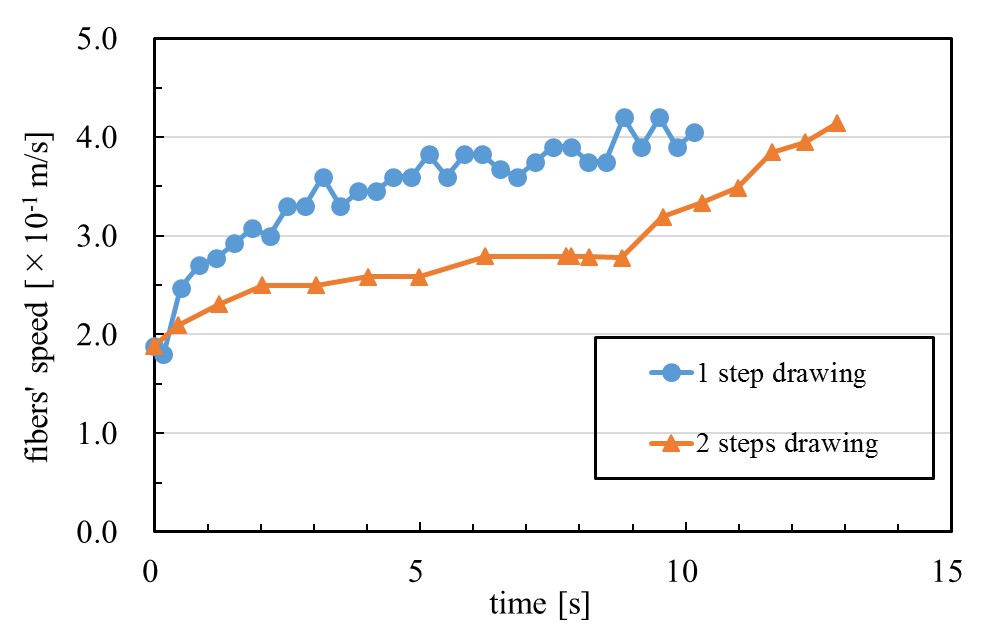
The behavior of drawing process was investigated by focusing on fiber stretching speed (spinning acceleration) to improve fibers' luster quality. Wet spinning method often has a problem of low luster level caused in higher spinning speed. We found that, (1) luster quality could be evaluated by fibers' arithmetic average (RΔa), which indicated fibers' roughness and (2) fibers' roughness was related to spinning acceleration by analyzing RΔa. Spinning acceleration was measured by chasing markers which was used to tie fibers. Regardless of length of spinning bath, fibers were stretched mostly during the first stretching stage. So we tried to stepwise drawing method. In case drawing ratio is 220 percent by one step, RΔa was 10.1 degree but by stepwise drawing (1st drawing ratio was 148.3 percent and 2nd drawing ratio was 148.3 percent) RΔa was decreased to 8.3 degree. We have cleared that stepwise drawing method has enabled to decrease fibers' roughness by preventing sudden change of fiber stress. In addition, we have cleared that higher temperature also improved fibers' roughness. In higher temperature, roughness was decreased in spite of higher acceleration because that fiber was easier to stretch than in lower temperature.
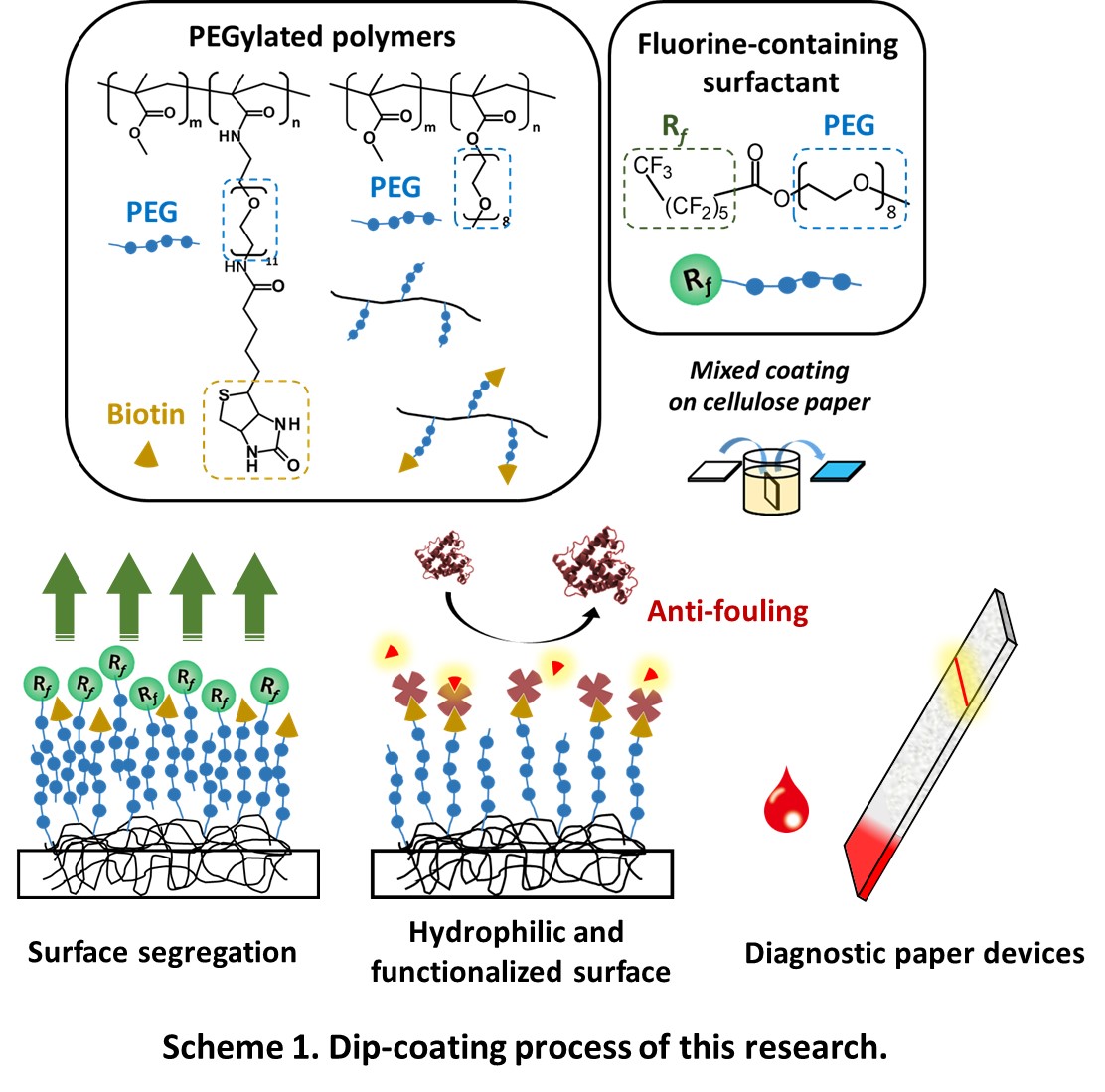
Paper-based devices have attracted a lot of attention as simple analytical platforms for bioassays. However, paper-based biosensors have still many problems, especially nonspecific protein adsorption. Detection sensitivity and selectivity of such sensors are impaired by nonspecific protein adsorption because of the drifting and denaturing of the detection ligands, the loss of target analyte during transport to the detection zone, and background noise from nonspecific protein adsorption. Typically, this problem in a sensor device is addressed by surface modification with hydrophilic polymers. For example, poly(ethylene glycol) (PEG) provides protein-repellent properties on wide variety of surfaces.
Herein we demonstrated surface modification of cellulose paper with a functional PEGylated polymer via simple dip-coating. This functional PEGylated polymer has biotin as a reactive site on the end PEG chains. In general, dip-coating with a PEGylated polymer followed by drying generates a hydrophobic surface because the hydrophobic moieties of the polymer are preferentially segregated at the air-polymer interface to minimize the surface energy. To display the hydrophilic moieties (functional parts) of a polymer on an outermost surface in dip-coating, we previously proposed that the surface segregation in the dip-coating of a PEGylated polymer can be controlled using a fluorine-containing surfactant. Based on the previous study, we here attempted to impart antifouling property and reactivity with other molecules such as antibody, DNA to the paper surface.
We synthesized PEGylated copolymers (Scheme 1) and a fluorine-containing surfactant (Rf-PEG). Cellulose paper was dip-coated with these copolymers in the presence of Rf-PEG. This coted paper had water absorbency in comparison with PMMA-coated paper and a paper coated PEGylated copolymers in the absence of Rf-PEG. Moreover, this coated paper had less nonspecific protein adsorption than a raw paper and a paper coated PEGylated copolymers in the absence of Rf-PEG. We expect that this coted paper will be applied to platforms for bioassays.
The excited electrons and holes generated upon by irradiating UV ray to TiO2 have powerful redox power. They produce reactive oxygen species (ROS) under aerobic conditions at the surface. These species rapidly decompose organic compounds to converts them into CO2 and H2O via carbon-carbon bond cleavage. TiO2 has been used as a powder to increase the catalytic effect. However, the use of particles involves the need of incorporating extra stages in the purification process, such as separation and catalyst recycling. We focused on a composite of TiO2 nanoparticles and polymer nanofibers. Polymer nanofibers have a large surface area. Polymer nanofibers are fabricated by electrospinning as follows. An electric field is used to create a charged jet of polymer solution. As this jet travels in air, the solvent evaporates leaving behind a charged fiber that can be collected on a metal plate. The purpose of this study is to develop novel composite polymer nanofibers with TiO2 nanoparticles and clarify its applicability as photocatalyst for degradation of organic compounds. N-isoprorylacrylamide-co-N-methylol acrylamide (NIPA-co-NMA) polymer was synthesized by free radical polymerization. NIPA polymer is a thermosensitive polymer, and NMA can be used as a crosslinking agent. The composite nanofibers were fabricated by electrospraying methanol containing NIPA-co-NMA polymer and TiO2 nanoparticles onto stainless-steel plate through a nozzle with application of high voltage. The composite nanofibers catalyzed decomposition reaction of methylene blue (MB) successfully.
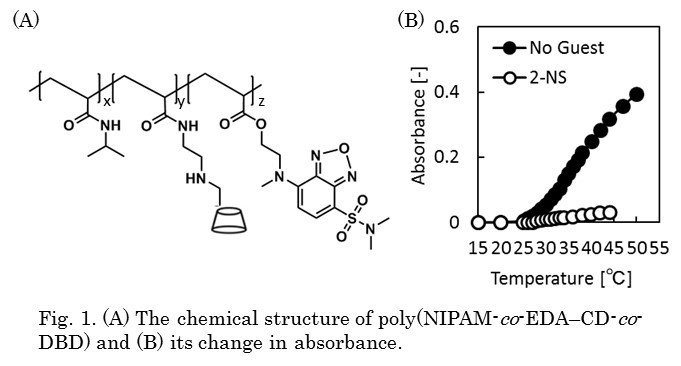
Development of novel materials which mimic the excellent molecular recognition function of biomembranes has recently got attention. Copolymer of N-isopropylacrylamide (NIPAM) bearing β-cyclodextrin (CD) (poly(NIPAM-co-CD)) exhibits molecular recognition function, which controls its volume phase transition behavior by the inclusion of guest molecules[1]. However, the mechanism of the molecular recognition of poly(NIPAM-co-CD) has not been fully explained. N,N-dimethylaminosulfonyl-2,3,3-benzoxadiazole (DBD) is a fluorophore and changes its fluorescence intensity in response to hydrophilicity around the molecule[2]. If DBD is introduced into the poly(NIPAM) chain, changes in polarity around PNIPAM accompanying phase transition can be detected.Herein, DBD is copolymerized with poly(NIPAM-co-CD) to directly analyze the change in hydrophilicity of the poly(NIPAM-co-CD) chains by inclusion of guest molecules (Fig. 1A). We discuss the correlation between the phase transition behavior and the hydration state around NIPAM, which is controlled by molecular recognition of CD in polymer chains. The change in phase transition and hydrophilicity around the polymer chains caused by inclusion of a guest molecule, sodium 2-naphthalenesulfonate (2-NS), was investigated by cloud point measurement and fluorescence intensity measurement. Fig. 1B. shows the results of the cloud point measurement. When 2-NS were added, aggregation of the polymer was suppressed. The fluorescence intensity measurement exhibited that there was no difference in the change in fluorescence intensity by addition of 2-NS, suggesting that inclusion of 2-NS in CD did not affect the hydrophilicity around NIPAM. Further, the cloud point measurement in NaCl solution did not display suppression of aggregation even the presence of 2-NS, resulting from electrostatic shielding by Na+ around SO3– group of 2-NS which was included in CD. It implies that the suppression of aggregation in poly(NIPAM-co-EDA-CD-co-DBD) by addition of 2-NS was caused by the electrostatic repulsion of 2-NS included in CD.
[1] Macromolecules, 45, 9742-9750, (2012)
[2] Anal. Chem.,, 75, 5926-5935, (2003)
To investigate the effect of the surface properties and polymer properties on surface modification via electrostatic adsorption, the electrostatic adsorption behavior of zwitterionic copolymer on negatively charged surface was studied. A series of positively charged zwitterionic copolymer and a series of negatively charged surfaces including porous substrates and dense films were respectively fabricated. The electrostatic adsorption behavior of zwitterionic copolymer on negatively charged porous substrates were confirmed by contact angles and fluorescently labelled protein adsorption experiment. And the adsorption behavior of zwitterionic copolymer on negatively charged dense films were confirmed by quartz crystal microbalance (QCM) determination and fluorescently labelled protein adsorption experiment. The results indicate that the lower charge density on the zwitterionic copolymer bring to the higher adsorption on the charged surface, while the extremely low charge density results in the lower adsorption on the charged surface, due to the low interaction. The high density of film surface charge is beneficial for surface adsorption, while the extremely high density of the film surface charge leads to the lower surface adsorption, due to the steric hindrance of the negatively charged sites. This work provide the insight into the best strategy for surface modification via electrostatic adsorption.
Gating membrane is a porous membrane that opens and closes pores responding to heat[1] and specific ions[2]. Recently sensor system using photo-responsive material attracts attention because of several advantages such as simple operation and uninfluential nature to the substance. In this research, we aim to develop a photo-responsive gating membrane that opens up the possibility of conventional membranes.
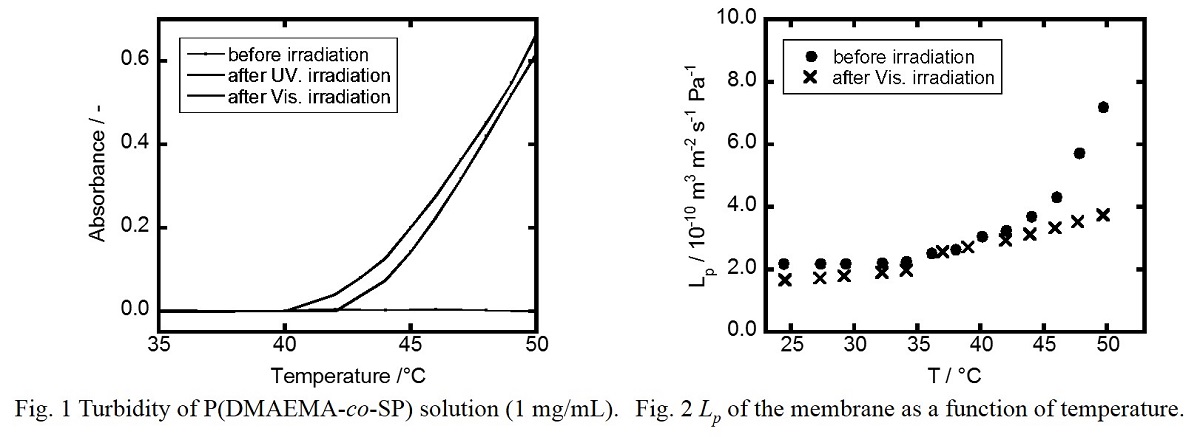
Photo-responsive polymer composed of poly(2-(dimethylamino)ethyl methacrylate) (PDMAEMA) actuator and photo-sensing spiropyran (SP) was synthesized by free radical polymerization (molecular weight : Mn =1.49 × 104 , Mw / Mn = 2.07 and SP content: 0.81 mol% ). Lower critical solution temperature (LCST) of 0.1 wt% aqueous solution is 42 °C before visible light (Vis) irradiation and 40 °C after Vis-irradiation while no LCST was observed after ultraviolet light (UV) irradiation.
The copolymer was immobilized to the polyethylene (PE) porous membrane by plasma-induced graft polymerization (Graft ratio: 5.6% and SP content: 0.96 mol% ). Permeation coefficient (Lp) of the prepared membrane increases as water temperature rises before light irradiation (Fig 2). This is because the copolymer shrinks above LCST and opens the pore of the membrane. Just after UV-irradiation, Lp of the membrane decreases as we expected, but increases then. The latter behavior may be due to elimination of the copolymer from PE substrate by UV-irradiation, as indicated by FT-IR spectral change. After Vis-irradiation, Lp of the membrane did not increase even though linear polymer shows similar LCST behavior before and after Vis-irradiation (Fig 1). This is probably because LCST behavior of grafted polymer in the pore and linear polymer in diluted solution is different.
In conclusion, we have successfully fabricated visible light-responsive gating membrane.
1) H. Kuroki et al., J. Membr. Sci. 2010, 352, 22-31.
2) T. Yamaguchi et al., JACS, 1999, 121, 4078-4079
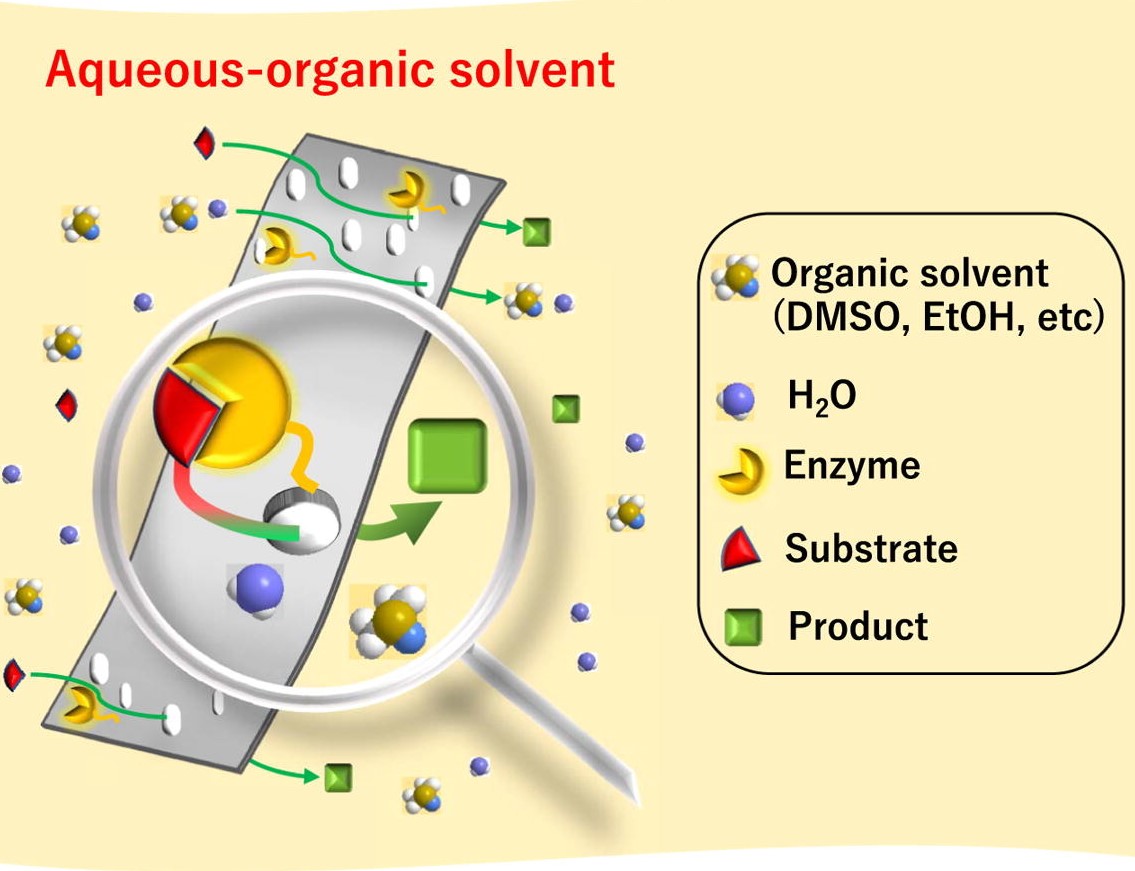
Enzymes play vital roles in biological transformations due to incomparable selectivity. Enzymatic membrane reactors (EMRs) combine enzymes with membranes, and many researchers have studied the synergistic effect of EMRs exerting on enzyme performance. Before the utility of EMRs can expand from natural aqueous media to organic solvents, robust membranes must be developed to promote enzyme protection from hostile forms of media. For this study, laccase was immobilized on an organic-solvent-resistant hydroxylated polyketone (PK-OH) membrane via covalent bonds and served as a model enzyme. Ketone groups facilitated the immobilization via hydrogen bonds, leading to a high immobilization density of 462 μg/cm2. In homogeneous aqueous-organic solvents, the activity of immobilized laccase was up to 3.5 times greater than that of free laccase towards 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid). In addition, the results also showed improved activity towards highly concentrated 2,4,6-trichlorophenol and bisphenol A (1.4-g/L). Furthermore, the activity in filtration mode showed a 240% increase over that in batch mode. The immobilized laccase maintained its activity after 40-days of storage, 10 reuse cycles, and 50 h of continuous reaction. These results show that robust polyketone based membrane support will create opportunities for the application of EMRs in aqueous-organic solvents.
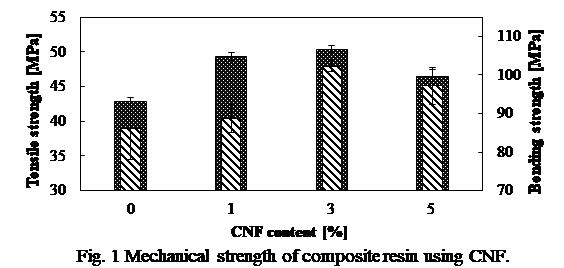
Cellulose nanofiber (CNF) is a micro-refined nano-level fibrous material that can be produced from chemical and mechanical treatment of variety of woody biomass materials. CNF has attractive characteristics, it's density is 5 times lesser than that of steel while being 5 times stronger. Moreover, it is produced from natural biomass resources therefore it is entirely environmentally friendly material1). Recently, its compounds with resins have received increased attention as alternative to pure metals owing to their light-weight and noncorrosive characteristics. In this use, defibrating and surface hydrophobization have main roles in obtaining high quality CNF. CNF is hydrophilic, while phenolic resins are hydrophobic; therefore, mixing them could result agglomeration and consequently lower the strength of the composite. In addition, the form of CNF is likely to influence the properties of the composites. Application of thermoplastic resins, such as polyethylene and polypropylene, in the formation of CNF composite materials have been widely reported. However, to the best of our knowledge, few studies have been conducted using thermosetting resins in the synthesis of CNF-reinforced composite materials. In this study, reinforcing of phenolic resin with CNF as a model for thermosetting resins was investigated. Various tests were conducted to evaluate the performance of the composites produced with CNF (produced at different physico-chemical conditions) in different proportions with the resin. Various techniques, such as Fourier-transform infrared spectroscopy, mechanical testing, and scanning electron microscopy were utilized to understand their properties. Fig. 1 shows mechanical strength of typical CNF composite. As shown, bending strength as well as tensile strength increased approximately 20% when 3 wt% CNF was used. This study suggested that reinforcement of the resin using CNF resulted in stronger and more promising composites.
References: 1) Akihiro Sato et al., Composites Part A: Applied Science and Manufacturing, 83, 72-79 (2016)
This study is to investigate the characteristics of polyvinyl alcohol (PVOH) added with different loading level of calcined fish bone (HAp). HAp was extracted from Chirocentrus nudus skeletal via thermal calcination; followed by solution casting method to produce PVOH composite films, consisting 0 phr to 5 phr calcined fish bone. In term of tensile strength, it decreases upon added with 1 phr calcined fish bone then increases at 2 phr loading level. The addition of 3 phr calcined fish bone was found to be the optimum loading level to exhibit reinforcement function; whereas for elongation at break, a decreasing trend with an increase in calcined fish bone loading level was found due to the continuity of chain mobility in PVOH being interrupted and the inducement of internal friction within PVOH matrix. For scanning electron microscopy, the wave-liked shrinkage, fibrils and agglomerates were reduced with increasing calcined fish bone loading level, indicating a high tensile strength and low elongation when increasing the loading level. In x-ray diffraction analysis, the peak broadening effect indicates a decrease in HAp crystallite size which has ruptured and well dispersed within PVOH matrix. For differential scanning calorimetry, the enthalpy of melting and crystallinity (in terms of qualitative and quantitative) possessed similar trend as the finding in tensile strength. In fourier transform infrared spectroscopy analysis, the unique peaks occurred approximately at 3263 cm-1 and 2923 cm-1, indicating the presence of O–H group bonding and C–H bonding respectively. Lastly, this study concluded that the 3 phr calcined fish bone was the optimum loading level to be employed in biomedical application such as bone scaffolding due to the high strength and compatibility.
Polymer/magnetic particle composites have high magnetic sensitivity, and thus have wide applications in biomedical fields. B. Tural et al. reported recently that if polymer containing a carboxyl group such as poly(methacrylic acid) was added to the coprecipitation solution during magnetite nanoparticle (MNP) synthesis, the polymer was immobilized on the magnetite surface immediately following particle generation. The method is one-step process that does not require complex and time-consuming manipulation.
However, so far, there are no reports on what kind of polymer (or functional group) can be used for this type of one step synthesis of polymer/MNP composite. Therefore, in this study, we tried one-step synthesis of polymer/MNP composite by using different polymers which contains different functional group to clarify 1) what kind of functional groups can be applicable for one step polymer/MNP synthesis, and 2) the effect of the polymer difference on the immobilized polymer amount.
In the experiment, the synthesis of polyacrylic acid (PAAc) / MNP was carried out using the following procedure; first, PAAc, iron (II) sulfate heptahydrate and iron (III) chloride hexahydrate were dissolved in deionized water. Then, sodium hydroxide was dropped into the solution at room temperature to proceed MNP synthesis reaction. Polyacrylamide (PAAm), polyethylene glycol (PEG), polyethylene imine (PEI) and polyvinyl alcohol (PVA) immobilized on MNP were also synthesized using the same procedure just by changing PAAc to the corresponding polymers. The results of XRD analysis showed that for all the polymers used in this study, formation of MNP was confirmed, and the fact indicated that the presence of the polymers did not affect on the synthesis of MNP. Thermogravimetric analysis (TGA) indicated that for all the polymers, polymer immobilized on MNP was successfully synthesized, and the amounts of the immobilized polymers were in the order of PEG < PEI < PAAm < PAAc.
To prevent aggregation and oxidation of magnetite nanoparticle (MNP), silica coating on MNP was proposed. Recently, Setyawan et al. successfully achieved simultaneous magnetite nanoparticle formation and the modification of its surface with silica layer by using the electrooxidation method. Advantage of the method is that the process is very simple and easy since it's a one-step process.
The purposes of this study are following; 1) to synthesize MNP with silica coating under various conditions by electrooxidation, and to examine the effect of the synthesis conditions on the characteristics of the resulting sample, 2) to immobilize thermoresponsive copolymer, poly(NIPAM-co-AA) on the MNP with silica coating, and to evaluate their immobilized copolymer amount and Cu adsorption property.
As a result of X-ray diffractometer (XRD) analysis and Fourier transform infrared spectroscopy (FT-IR) measurement, it was confirmed that MNP with silica coating was successfully synthesized using this method. When the current value was changed during electrooxidation synthesis, it was found that the minimum particle diameter(maximum specific surface area) was obtained at the current value of 0.60 A. The results of thermogravimetric analysis (TGA) showed that the maximum immobilized amount of the copolymer of 0.255 g/g-MNP was obtained when the sample synthesized at a current value of 0.60 A was used. The immobilized amount was 3 times larger than that of the sample prepared using MNP without silica coating. This is considered to be due to less aggregation of MNP during polymer immobilization. The results of Cu (II) adsorption experiment showed that although Cu (II) recovery amount through temperature swing was similar to those of conventional thermoresponsive “gel” type heavy metal adsorbents, time required for Cu(II) recycling process could be decreased to 1/10. These results showed the effectiveness of this method.
Graphene-based materials have been studied for prospective applications in many fields, including catalysts and electrode materials. Graphene-based materials can be made by two kinds of approaches. The first one is the bottom-up approach represented by a chemical vapor deposition method. The second one is the top-down approach via graphene oxides. Although the latter approach is suited for mass production, it is difficult to control the crystallinity and the amount of defects by this approach. Therefore, tailoring some properties depending on applications has remained a significant challenge. We reported that it is possible to control the crystallinity and the concentration of defects by microwave-assisted (MW-assisted) annealing for carbon materials by adjusting distances between carbon particles through changing the bulk density. However, it can be difficult to change the bulk density depending on some types of carbon materials. This study is aimed at controlling the crystallinity and the concentration of defects regardless of the kind of carbon materials through development of a new up-flow reactor for MW-assisted annealing. Crystallinity as well as the concentration of defects were investigated by multiple techniques including Raman spectroscopy and thermogravimetric analysis. During MW irradiation, we observed moderate fluidization of sample particles in an up-flow reactor. Therefore, distances between carbon particles were adjusted, through which the concentration of defects was controlled. Our results indicate that extend of changing of the concentration of defects depends on the sample particle size and the gas flow rate during MW irradiation. Moreover, samples annealed by the up-flow reactor exhibited high oxidation resistance in air in comparison with ones treated by a down-flow reactor.
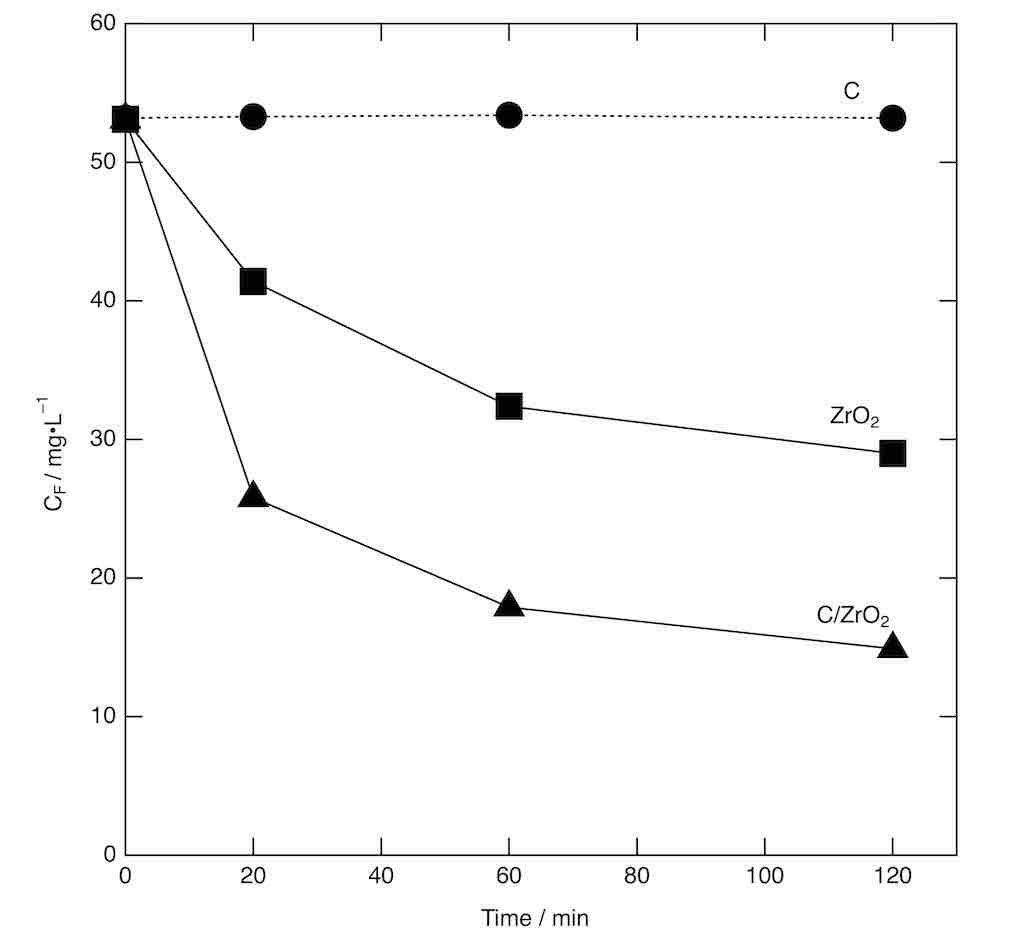
Magnesium oxide (MgO)-templated mesoporous carbons were modified with hydrated zirconium oxide (ZrO2·xH2O) synthesized through hydrolysis of zirconium chloride oxide (ZrOCl2) to prepare adsorbents for fluoride ion (F-) removal from water. Powder X-ray diffractometry (XRD) revealed that ZrO2·xH2O was successfully formed on the surface of mesoporous carbons. Adsorbed amount of F– was observed as a function of time by using pristine mesoporous carbons, modified mesoporous carbons, and ZrO2·xH2O prepared without carbon. The figure shows the F– concentration of solution (CF) as a function of contact time. The pristine carbon was found to adsorb little amount of F–. In contrast, the modified carbon and ZrO2·xH2O were capable of removing F– from water. The amount of F– removed with the modified carbons was larger than that observed using ZrO2·xH2O. This result indicates that the capability of ZrO2·xH2O to remove F– is improved by attaching it to the mesoporous carbon surface. The authors presume that this positive effect is probably provided by formation of ZrO2·xH2O on the carbon surface giving crystals that are smaller than crystals fomed without carbon. The F– removal was described by conventional adsorption isotherms. The other results such as effect of mesopore size and pH on the F– removal will be shown at the conference.
Porous carbon materials have attracted much attention because of their large surface area, large pore volume, chemical inertness, and electrical conducting properties. Mesoporous carbons with well-tailored pore system are essential for a number of applications, such as adsorbents, catalyst supports, and energy storage devices. Recently, a great deal of effort has been made to develop a method of synthesizing mesoporous carbons, including hard- and soft-templating techniques. Hard-templating techniques require preparation of inorganic template such as mesoporous silicas, impregnation of pores with appropriate carbon precursors, carbonization, and subsequent selective removal of inorganic template using HF
or NaOH. Another recently developed approach is based on the application of soft-template such as triblock copolymers access for simpler and facile synthesis of mesoporous carbons because of the avoidance of multiple step to prepare and remove the hard-template. However, the soft-templating techniques still suffer from long preparation durations and the excessive use of solvents for the formation of mesostructure in solutions through the self-assembly process, which causes low efficiency, high production cost, and environmental burden.
Herein, we investigate a highly efficient and simple approach to synthesize mesoporous carbons using solid raw materials under solvent-free conditions. The solid–solid approach has potential advantages for large-scale production, low cost, environmental-friendly process. Mesoporous carbons were obtained by the solvent-free technique using various combinations of solid raw materials with adjustments of the molar ratio.
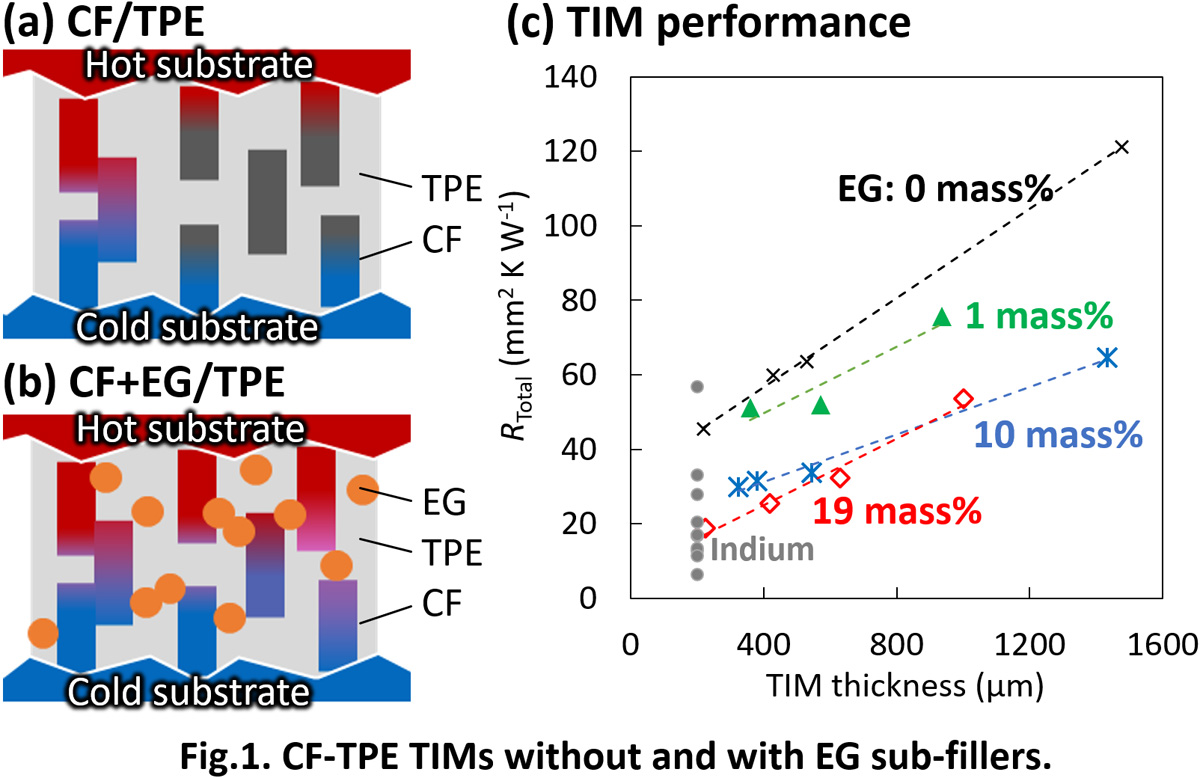
The demand for high-performance thermal interface materials (TIMs), which enhances releasing heat from the electronic devices, is increasing for further performance improvement and miniaturization of electronic devices. The performance of TIM is characterized by the total thermal resistance RTotal which is the sum of the bulk thermal resistance and the contact thermal resistance between the TIM and the opposing substrate. Therefore, it is required for the TIM to have high thermal conductivity for vertical direction κ⊥ and softness, thus conventional TIMs consist of filler particles having high thermal conductivity and soft polymer matrix. By using fillers with high aspect ratio and aligning them vertically, κ⊥ can be improved [1]. We previously fabricated TIM using carbon fiber (CF) having high thermal conductivity and high aspect ratio. By a simple process using shear force, CFs were filled in the thermoplastic elastomer (TPE) matrix with vertical alignment and high mass ratio of over 50 mass% (Fig. 1a) [2]. However, the total thermal resistance is as high as 100 mm2 K W-1 possibly because of the poor connections between the CFs and between the CF and the opposing substrate surface. This time, expanded graphite (EG) having high thermal conductivity and flexible sheet-like structure is added as a sub-filler (Fig. 1b). Thermal resistance was measured by the steady-state method and plotted against the TIM thickness (Fig. 1c). The thermal resistance decreased with the EG content, resulting in the significant reduction of the thermal resistance to RTotal = 19 mm2 K W-1 with 19 mass% EG. This value is similarly small as the high-performance TIM of an indium sheet. The improvement comes from both the improvement of the thermal conductivity (slope) and the reduction of contact thermal resistance (intercept).
[1] K. Uetani, et al., Adv. Mater., 26, 5857 (2014).
[2] R. Yamada et al., The 51st FNTG symposium, 2P-38 (2016).
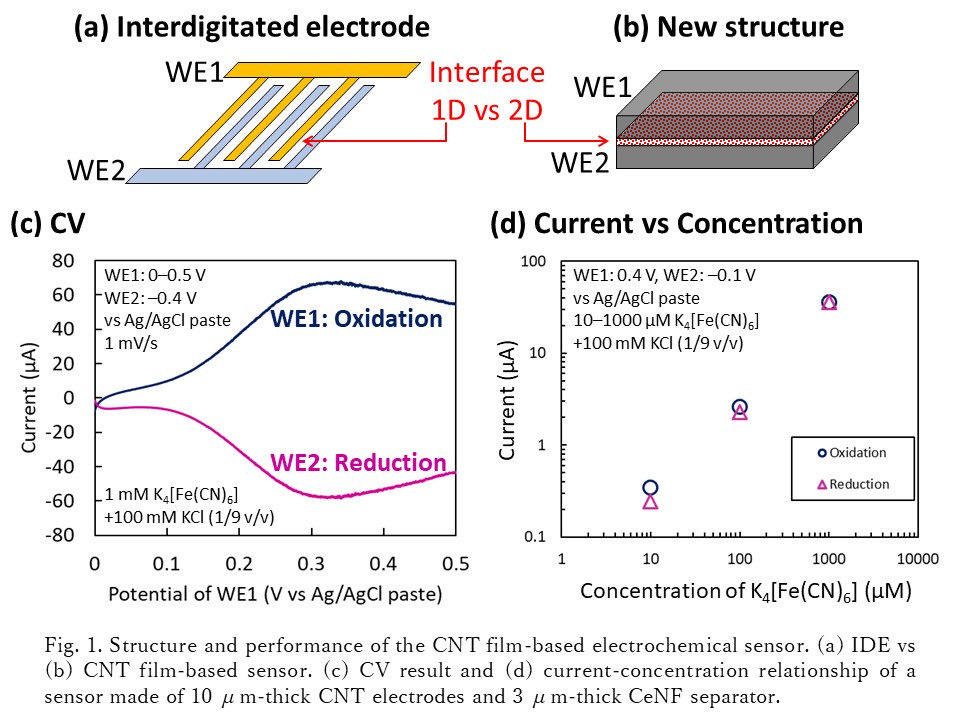
Electrochemical sensor based on redox cycle of repeated oxidation and reduction has advantages of compactness and high sensitivity. To promote the redox cycle, two working electrodes for oxidation and reduction should have large surface area, be placed closely, be insulated electronically while be conductive ionically each other. The typical example is the interdigitated electrode (IDE) (Fig. 1a), but the fabrication of the microstructure requires complex processes using lithography. In this work, we propose a new structure similar to electrochemical capacitor in which a separator made of cellulose nanofiber (CeNF) paper holds two working electrodes (WEs) made of carbon nanotube (CNT) paper (Fig. 1b). Cyclic voltammetry (CV) using K4[Fe(CN)6] aqueous solution shows the effectivity of this structure as a sensor (Fig. 1c).
Larger current was obtained with 3D electrodes of 10 μm-thick CNT papers and separator of 3 μm-thin CeNF paper, enabling detection of K4[Fe(CN)6] for concentrations down to 10 μM.
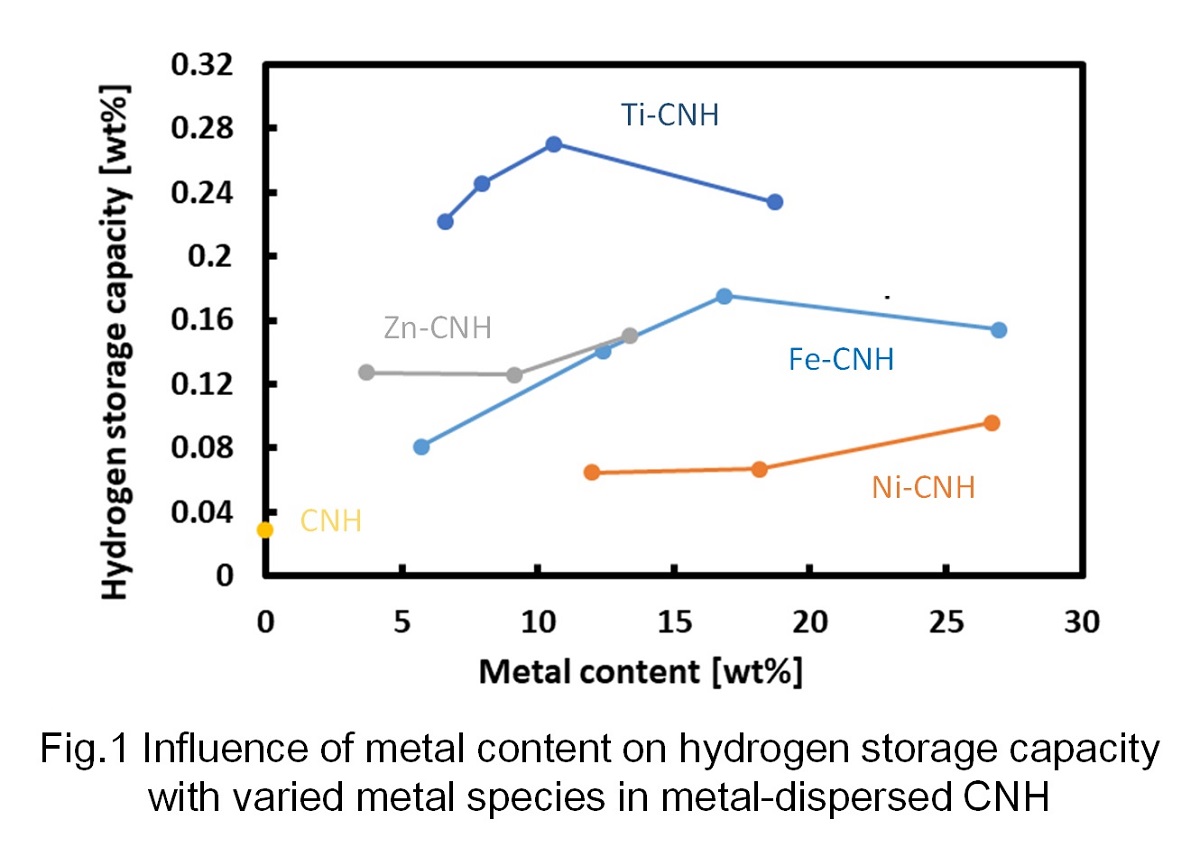
The conventional hydrogen storage materials such as metallic alloys are heavy and difficult to transport, and there is a problem that their costs are high. Therefore, in this research, we aim to develop hydrogen storage materials using carbon nanohorn (CNH), which is a lightweight and inexpensive carbon material.
CNH dispersed with metal nanoparticles was prepared using arc-in-water method. After that, the amount of absorbed hydrogen was measured under the condition of 0.95 MPa and 25 °C, and the influence of the metal composition and the content on the amount of absorbed hydrogen was clarified. It can be seen in Fig. 1 that there is the optimum metal content to maximize the hydrogen storage capacity. The increase in the amount of hydrogen storage with the increase in the metal content in relatively low metal content range is considered to be due to the increase of hydrogen spillover effect (dissociative adorption of H2 on carbon surface close to metal nanoparticles). Further, the decrease in the storage amount with metal content in high metal content range is considered to be caused by the decrease in carbon surface per material. Also, it can be observed that the amount of hydrogen storage becomes the highest when using Ti to disperse in CNH among Fe, Ni, Zn and Ti.
In addition, semi-empirical molecular orbital calculation was conducted in order to theoretically verify the hydrogen spillover phenomenon on the metal dispersed CNH. As a result, the hydrogen spillover effect is likely to occur at high pressure. It was shown that the hydrogen spillover effect can easily occur when H2 molecules present at the interface between Fe cluster and C atoms.
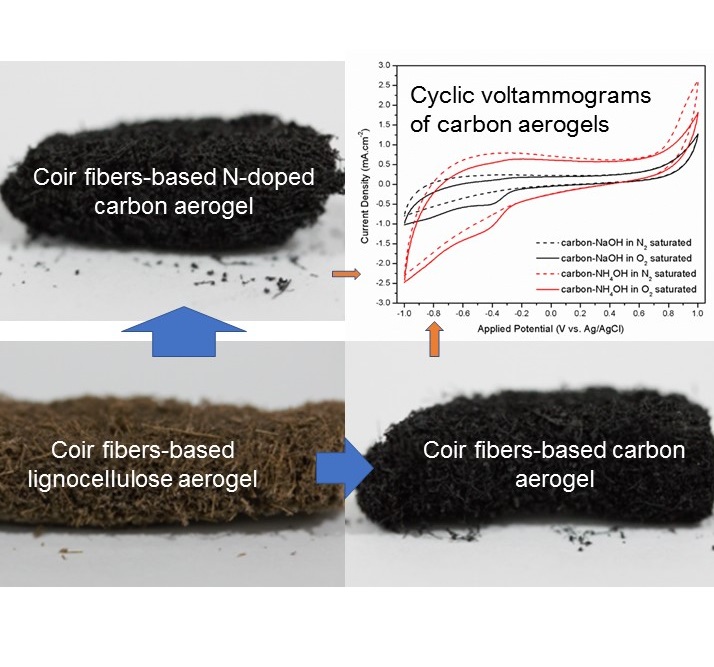
Metal-air battery and fuel cell are widely considered a promising candidate for next-generation renewable energy. One of the main problems of these technology is the catalyst for oxygen reduction reaction (ORR) that mainly depending on Pt, a very expensive material. The oxides of several transition metals have also been proposed for the purpose. Here, we propose nitrogen-doped carbon aerogel from coir fibers as an alternative metal-free electrocatalyst for the ORR. The nitrogen-doped carbon aerogel was prepared from coir fibers via simple ammonia–urea method followed by freeze drying and pyrolysis.Nitrogen molecules also have been well doped into the carbon structure, as confirmed by the existence of C=N functional group in FTIR spectra. Ammonia treatment could exfoliate the carbon structure causing defects and disorders, enhance the specific surface area, and maintain the morphology of carbon aerogel during carbonization. Kouteckỳ-Levich plots presented that the obtained nitrogen-doped carbon aerogel involved n ≈ 2 electrons towards ORR. The results suggest that the prepared nitrogen-doped carbon aerogel is a promising metal-free electrocatalyst toward ORR through 2e- pathways.
Proton-exchange membrane fuel cells, which can provide clean and efficient electricity, contain precious Pt electrocatalysts due to the sluggish kinetics of the oxygen reduction reaction (ORR). One of the major strategies to enhance the ORR catalytic activity is to control the morphology of the catalytic particles. Herein, a facile shape-directing mechanism was developed, and highly active Pt-based electrocatalysts were synthesized. Moreover, ammonium hexachroloplatinate(IV), which is known as recycling intermediate, was used as metal precursor of the synthesis experiments. It was revealed that NH4+ ions can behave as shape-directing agents in the presence of Cl– ions and O2. Based on the mechanism, Pt nanoparticles were synthesized in aqueous NH4Cl solution, forming a plate-like morphology that selectively exposes the {111} facets. Then, ammonium hexachroloplatinate(IV) and Cu salts were reduced together under the same synthesis condition, leading to Cu-doped Pt nanoplates. The Cu-doped Pt naonplates exhibited 3.7-times higher ORR catalytic activity than the commercial Pt catalysts. The present study, in which recycling intermediate has been used as metal precursor of highly active electrocatalysts, suggests a possibility to interconnect between material synthesis and recycling process.
Polymer electrolyte fuel cells (PEFCs) have received a great deal of attention for its utility in various applications, owing to their high energy conversion efficiency and clean emissions. However, a sluggish oxygen reduction reaction (ORR) rate and the low durability of conventional catalysts in PEFCs limits the wider commercialization prospects. To address these issues, our group had developed a carbon-free, connected platinum–iron (Pt–Fe) nanoparticle catalyst having porous hollow capsule structure. This catalyst exhibited an enhanced ORR activity as well as excellent durability against start-stop operations, due to the carbon-free structure. [1] Thus, the connected nanoparticle catalysts are identified promising, and further improvement in the ORR activity and durability can cater to the growing demands for PEFCs.
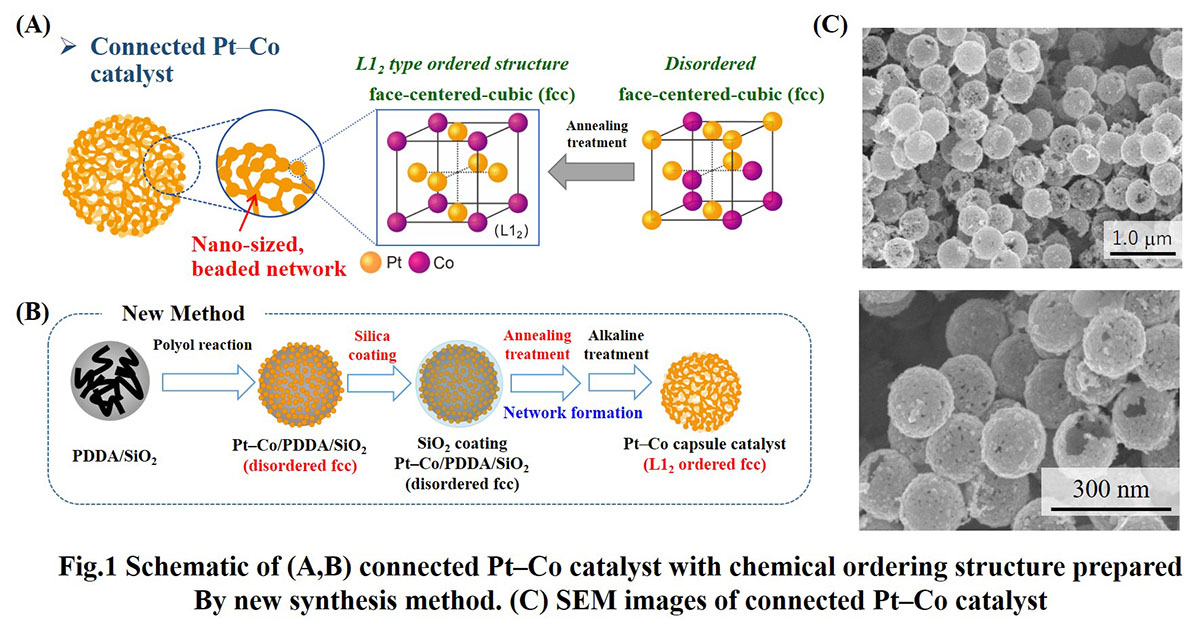
In this work, we developed a new connected catalyst, platinum–cobalt (Pt–Co) with a chemically ordered structure (FIG. 1A) having different catalyst atomic structures to that of connected Pt–Fe. Herein, we employed a new synthesis method for the catalyst processing, having the combination of silica coating and high temperature annealing as shown in Fig.1B. The SEM images (FIG. 1C) of the obtained Pt–Co catalysts showed a hollow capsule structure, indicating the formation of a connected Pt–Co network. The XRD patterns of the catalysts showed the peaks corresponding to an L12 type chemically ordered structure, when the annealing temperature was higher than 500 °C. Additionally, chemically ordered degree in the connected Pt–Co catalysts was successfully controlled by annealing temperature and time. Thus, we succeeded in the development and structural control of a connected Pt–Co catalyst composition for the first time. Investigating structural effects of connected Pt–Co catalysts on the ORR activity and durability is expected to provide useful guidelines to design an advanced connected nanoparticle catalyst for application in PEFCs.
References
[1] T. Tamaki et al., Energy Environ. Sci., 8, 2015, 3545–3549.
Polymer electrolyte fuel cells (PEFCs) have attracted much attention as promising energy conversion electrochemical devices. However, a conventional cathode catalyst of platinum nanoparticles supported on carbon black (Pt/C) exhibits a sluggish oxygen reduction reaction (ORR) rate and a low durability against start–stop and load cycle operations. In our group, a carbon-free, connected platinum–iron (Pt–Fe) nanoparticle catalyst with porous hollow capsule structure (FIG. 1A) has been successfully developed for an ORR. [1] This catalyst provides enhanced ORR activity and excellent durability during start-stop operations. However, there is a dissolution of alloyed metal occurring during load cycles, resulting in reduced ORR activity for the connected Pt–Fe catalyst.
In this study, we focused on a chemically ordered face-centered-tetragonal (fct) structure (FIG. 1A) to improve the load cycle durability of the Pt–Fe catalyst. The conventional synthesis method of connected Pt–Fe catalyst using a supercritical fluid treatment produces low fct degree (40–50%). Here, we propose a new and simple synthesis method using the combination of silica coating and high temperature annealing as shown in FIG. 1B. Using this method, we succeeded in preparing connected Pt–Fe catalysts having nano-sized network and a high fct degree (70–80%). The obtained catalyst exhibited a ten-times higher ORR specific activity, compared with that of the Pt/C. Furthermore, as showed in FIG. 1C, the connected Pt–Fe catalyst with the high fct degree (74%) exhibited a higher retention of ORR activity after 10,000 load cycles, compared to the low fct catalyst (44%). Thus, this study demonstrated that a highly ordered fct structure greatly contributes to the suppression of dissolution of metal, resulting in an improved load cycle durability of the connected Pt–Fe catalyst.
Reference
[1] T. Tamaki et al., Energy Environ. Sci., 8, 2015, 3545–3549.

We present an electrochemical reaction system that enables the generation of electricity, employing waste plastics as raw materials. These plastics can include polyvinyl alcohol, polyurethane, nylon, vinylon and polyethylene terephthalate. In the proposed process, the polymers are partially or completely dissolved in phosphoric acid (H3PO4) at temperatures of 100 °C or higher. When a physical mixture of plastic solid and H3PO4 solution was supplied to the anode in a temperature-controlled fuel cell, the in situ dissolved plastic component was oxidized to CO2, protons and electrons as a result of anodic polarization. This oxidation reaction at the anode was optimized by designing a catalyst support with an enlarged, ordered pore structure. An active oxygen species, believed to be the OH· radical, also plays a key role in the plastic oxidation reaction. The fuel cell demonstrated in this work functioned continuously to generate power densities on the order of mW cm-2(Fig.1). Conventional plastic disposal systems based on combustion emit toxic pollutants. The present study demonstrates a process that overcomes this issue by allowing the treatment temperature to be significantly decreased.
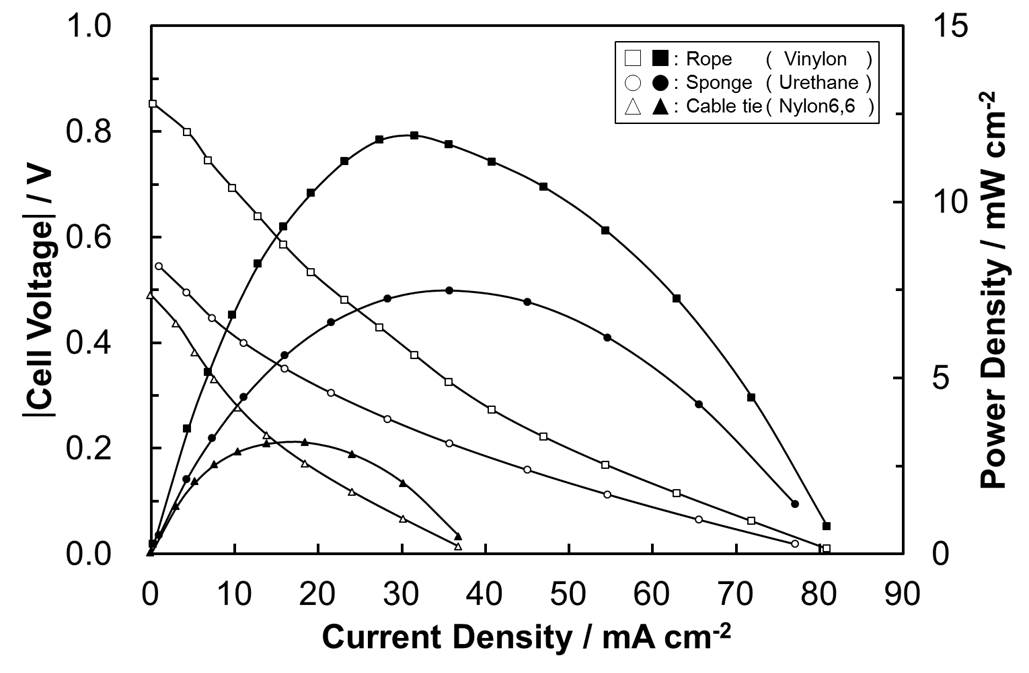
Transparent conductive oxides (TCO) with high visible transmittance and low resistance have been used in applications of optoelectronics. Dielectric/Ag/dielectric (DAD) multilayer was proposed to meet the requirements of low resistance and enough transparency [1]. In this work, a Cu-Ag bi-layer was proposed to serve as the mid-layer in oxides, including TiOx and WO3. All the films were prepared through E-beam evaporation at room temperature. The optical and structure properties of the D/Ag/Cu/D were studied by X-ray diffraction, UV-Vis, SEM, TEM and XPS. The surface morphology of Ag will be flattened by the Cu buffer layer, which can reduce the critical thickness of Ag layer. The thickness effects of Ag and Cu film on the optical properties of the D1/Ag/Cu/D2 were also investigated. According to the UV/VIS transmission spectra, the TiOx (35 nm)/Ag (12 nm)/Cu (3 nm)/TiOx (35 nm) exhibited the highest average visible light transmittance of 88%. The figure of merit (FOM) of TACT stacking is ~ 126 mホゥ-1 at the wavelength of 550 nm. Using WO3 with an thickness of 40 nm as the capping layer above Ag/Cu/WO3 (WACW) shows a FOM of 73 mホゥ-1. Use of Ag/Cu bilayer can widen the transmittance of the D1/Ag/Cu/D2 with an appropriate Ag and/or Cu thickness. The experimental results were also compared with the optical simulation to figure out the role of the ultra-thin Cu buried layer.
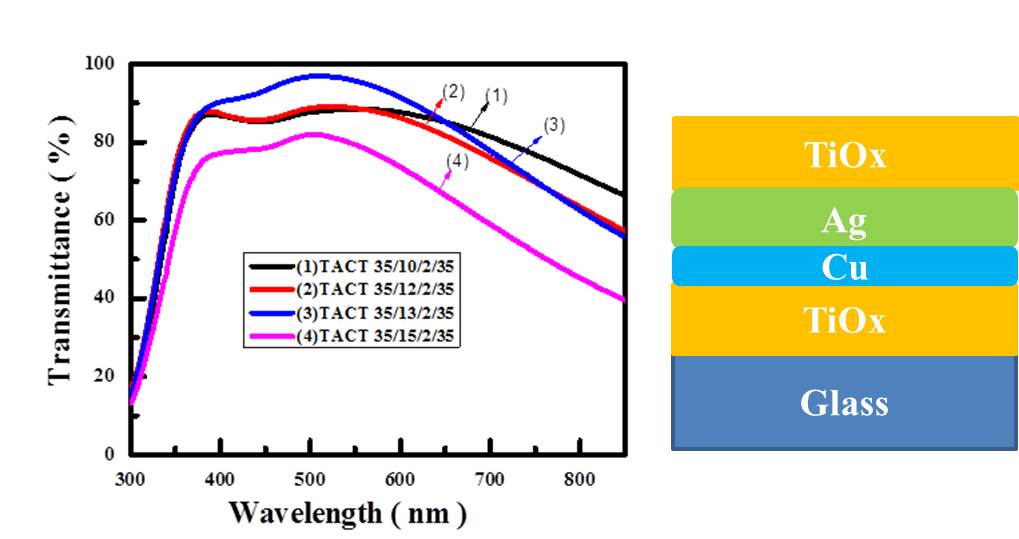
As chemical industries have grown gradually, strong alkaline chemicals are commonly used in almost all industries worldwide. Despite their usefulness, alkaline chemicals can be very dangerous as they can cause severe intoxication. In particular, gaseous alkaline chemicals require more precautions because they spread very quickly without being noticed in case of leakage. Therefore, it is important to detect strong alkaline chemical leaks promptly and easily. In this study, to detect gaseous alkalis immediately with naked eyes, we fabricated colorimetric textile sensors based on halochromic dyes. Three halochromic dyes with superior pH sensitivity were synthesized and applied to textile surface through screen printing method. The sensitivity to gaseous alkaline chemicals of the fabricated textile sensors was investigated. Ammonia was used as a gaseous alkaline chemical and the test was conducted at a concentration of 1 to 100 ppm. All of the fabricated sensors showed a high reaction rate and distinctive color change under alkaline condition due to the high pH sensitivity of the dyes. These results indicate that the fabricated textile sensors can be a promising candidate for practical sensor applications.
Recently, nanosheets (NSs) have attracted attention because of their various fascinating properties. We have developed the synthesis method of NSs inside bilayers of hyperswollen lyotropic lamellar phases of amphiphile solutions; the distance of the bilayers with several nm thickness is kept several hundred nm. We named the synthesis methods "Two-dimensional Reactors in Amphiphilic Phases" (TRAP) method. We have already successfully synthesized NSs of polystyrene and metal organic frameworks (MOF) in the thin hydrophobic sites inside the bilayers of aqueous amphiphilic solutions. To extend the range of the application of the TRAP method, we focused on aluminosilicate that is commonly synthesized in the hydrophilic reaction fields.
Aluminosilicate can be a precursor of zeolites with molecular-sized pores (0.3-1.2 nm-sized pores), which lead to the unique functions of zeolites: catalysts, adsorbents, ion-exchangers and membranes. CHA-type zeolites (CZs) are preferable catalysts for methanol to olefins (MTO) processes, which are important for the production of ethylene and propylene requied for polymer industry. CZs, however, have some problems; in particular, the pores are quickly clogged with by-products, and the resulting restriction of the reaction site and the diffusion induces the catalyst deactivation. One of the most promising solutions of the problem is the downsizing of the catalysts. Because the downsizing effect is based on the decrease of diffusion path length, CZ-NSs should have a potential as catalysts with one of the ideal shapes for the MTO processes.
Here, we report the preparation of the suspension of aluminosilicate NSs as a precursor to synthesize CZ-NSs using the TRAP method. Atomic force microscopy (AFM) and transmission electron microscopy (TEM) of the obtained CZ-NSs indicate that their thickness and horizontal width are a few nm and several hundreds of nm, respectively (Figure). This is the first example of CZ-NSs with a few nm thickness.
Seawater is currently used for desalination and salt production, and then the bittern is discharged by the operation. The bittern contains various resources, but most of them are unutilized. It is important to consider the resource recovery process from the bittern in Japan. Therefore, we focused on synthesis of layered double hydroxides (LDH) from the components of the bittern. The LDH is a clay mineral and has anion exchange ability. In this study, the anion exchange property of LDH from the bittern was investigated in comparison with that from ingredients of LDH stoichiometric concentration without components of Na, K, etc which include in the bittern but not in the basic LDH.
The simulated bittern was adjusted based on the actual composition of the bittern and was mixed with Al solution. The precipitate of LDH obtained by dropping into sodium hydroxide solution with coprecipitation method at a high pH region and the powder samples were obtained by filtration from the slurry. The structure, size, shape and ion concentration and anion exchange property of the LDH were analyzed by XRD, SEM and ICP.
The precipitated LDH from the bittern was Mg-type hydrotalcite as results of the XRD measurements. Then, the synthesis of Ca-type hydrocalumite can be also synthesized at another higher pH region. In the syntheses, all LDHs had Chloride ions as charged intercalants for keeping charge neutrality in the layer structures. The anion exchange property was investigated between Cl- and PO43-. The exchange structures were checked by interlayer distance variation before and after the exchange operation. The anion exchange capacity of the hydrotalcite form the bittern showed lower than that from the artificial ingredients.
Zeolites are crystalline microporous aluminosilicates that are widely used as adsorbents, ion-exchangers, and catalysts owing to their large micropore volumes with well-defined pore structures, ion-exchange abilities, and solid acidities. However, the pore sizes of zeolites are usually smaller than 0.7 nm, and the diffusion of molecules in the micropores is often a rate-determining step. Therefore, nano-sized zeolites attract much attention. Proper supports should be used for practical use of nanosized zeolites to prevent their aggregation, and porous inorganic submicron fibers are desirable supports for nano-sized zeolites due to strength, high formability and high surface areas. We prepared zeolite-containing submicron-fibers by centrifugal spinning. The centrifugal spinning is a method using a centrifugal force as a driving force and was developed recently for mass production of various fibers. After careful tuning of preparation conditions and fiber compositions, obtained fiber showed high BET surface areas even after repeated water adsorption test.
To meet increasingly strict regulations on emission control, there has been a high demand on effective catalysts for ammonia selective catalytic reduction (NH3-SCR) of NOx. Although the CHA zeolite dominates the current NH3-SCR market, other small pore zeolites have been studied as equivalent or more efficient catalysts. AFX zeolite is considered to be a superior catalyst for this reaction, which has not yet been applied for industrial use. This situation is in part due to partly because of its long synthesis time (typically, one week or even longer). A more efficient synthesis can help greatly save cost and energy which will pave the way for its industrialization towards emission control. In the work, we demonstrate the ultrafast synthesis of AFX zeolite from the hydrothermal conversion of FAU zeolite, which resulted in fully crystalline product in as short as 12 min.
We carried out the synthesis in the autoclave with a typical recipe, which took 7 d to yield AFX zeolite. The ultrafast synthesis was carried out in a tubular reactor made of stainless steel tube (1/4 inch) which features fast heating and cooling. To prompt the crystallization, seeding method was applied where the autoclave product was used as a seed; the composition of the synthesis precursor was optimized where FAU zeolite was used as a raw material; and, the synthesis temperature was elevated to 210 °C. Although the crystallization time was shortened, the synthesis resulted in the co-crystallization of ANA zeolite as a byproduct. To overcome this issue, the AFX seed was found to play an important role. With a seed synthesized by an improved method, the pure AFX zeolite was obtained after a synthesis of 12 min. This ultrafast synthesis method would facilitate the mass production of AFX zeolite boost its application as a de NOx catalyst.
The synthesis of zeolites, typically carried out in batch reactors like autoclaves, takes a time so long (typically, on the order of days) that the crystallization of zeolites had been believed to be very slow in nature. Long periods of hydrothermal treatment also cause a burden on both energy efficiency and operational costs. Recently, we have reported the ultrafast syntheses of a class of industrially important zeolites within several minutes. We present herein a continuous flow method for the synthesis of ZSM-5 using pressurized hot water with extremely high temperature (370 °C) as the heating medium. With a carefully tuned gel, the crystallization could be completed in tens of or even several seconds.
Direct mixing of synthesis precursor and the pressurized hot water in a millimeter-sized continuous flow reactor could result in immediate heating up to high temperatures; consequently, the crystallization of ZSM-5, from amorphous state to full crystallinity, proceeded to completion at a remarkably fast rate in a system without the addition of any seed. The well-tuned synthesis precursor, obtained by aging the initial aluminosilicate gel at 90 °C for a certain period, triggered the nucleation and ensured the formation of ZSM-5 without any byproduct at extremely high temperatures. The crystallization rate surpassed the decomposition rate of OSDA because of the creation of a favorable environment for crystallization. When a gel aged for 16 h was employed, fully crystalline ZSM-5 was obtained after a synthesis for 6 s. SEM image shows that the ZSM-5 synthesized after 6 s exhibited well crystallized facets. These unexpectedly fast crystallization rates in a continuous flow apparatus helped us to approach the limit of the zeolite crystallization.
Graphene oxide (GO) contains abundant oxygen-containing functional groups, which is an effective adsorbent to remove organic and inorganic pollutants. SBA-15 possesses large surface area, large pore volume, adjustable pore size, and thick silica wall. In the current study, we attempted to synthesize GO/SBA-15 nanocomposite. The large surface area of SBA-15 can provide abundant adsorption sites and plays a crucial role in increasing the adsorption efficiency and reuse of GO. This investigation characterized GO/SBA-15 materials through transmission electron microscopy (TEM), field–emission scanning electron microscopy (FESEM), X-ray diffractometry (XRD), surface area analysis, and Raman spectrometry. Various experimental parameters including the initial concentration of dye, calcination temperature, and pH of the solution were investigated. Immersion of GO into mesoporous SBA-15 increased methylene blue (MB) adsorption capacity. Increasing the initial concentration and solution pH enhanced the adsorption efficiency of GO/SBA-15. Optimal MB adsorption was observed at a calcination temperature of 550 °C. N2 adsorption-desorption measurement indicated that the GO/SBA-15 samples were mesoporous materials with uniform pore distribution. The addition of GO did not affect the mesostructure of the SBA-15 framework. The GO/SBA-15 possessed a large surface area of 891 m2/g, large pore volume of 1.035 cm3/g, and wide pore diameter of 6.50 nm. TEM of the synthesized GO/SBA-15 revealed that GO was homogeneously coated on the surface of mesoporous SBA-15. The adsorption isotherm and kinetics were more closely represented by the Langmuir model and pseudo-second order model, respectively, indicating that the adsorption type was monolayer adsorption. Reutilization experiments were carried out to investigate the stability and regeneration ability of the GO/SBA-15 materials. The adsorption capacity still remained above 95 % after five-time reused, demonstrating the excellent regeneration of the GO/SBA-15 materials. This method of preparing GO/SBA-15 composite is potentially useful for removing organic pollutants from wastewater.
Polycarbonates used in engineering are strong, tough materials, and some grades are optically transparent. Nowadays, the flame retardancy of polymers are the major concern. In this study, Polycarbonate (PC) was selected to develop an efficient flame retardant (FR) of high temperature polymer. A series of organo phosphorus flame retardants (FRs) based on aromatic phosphate and cyclic phosphate, mainly Biphenyl bis(cyclic 1,3-propanediol phosphate)(BP-CPP) and Biphenol bis(cyclic 2,2-dimethyl-1,3-propanediol phosphate)(BP-DMPP), were synthesized for polycarbonate. Their thermal stability and flame retarding efficiency as a single-component additive were investigated and compared with resorcinol bis(diphenyl phosphate) (RDP). Flame retarding efficiency was evaluated by the UL-94 test method. The V-0 rating was achieved at RDP/ BP-CPP loading of 6/2 wt% for polycarbonate, which is far better than that of resorcinol bis(diphenyl phosphate) and cyclic phosphate-based FRs. And its flame retarding performance was studied by the UL-94 and thermogravimetric analysis (TGA).
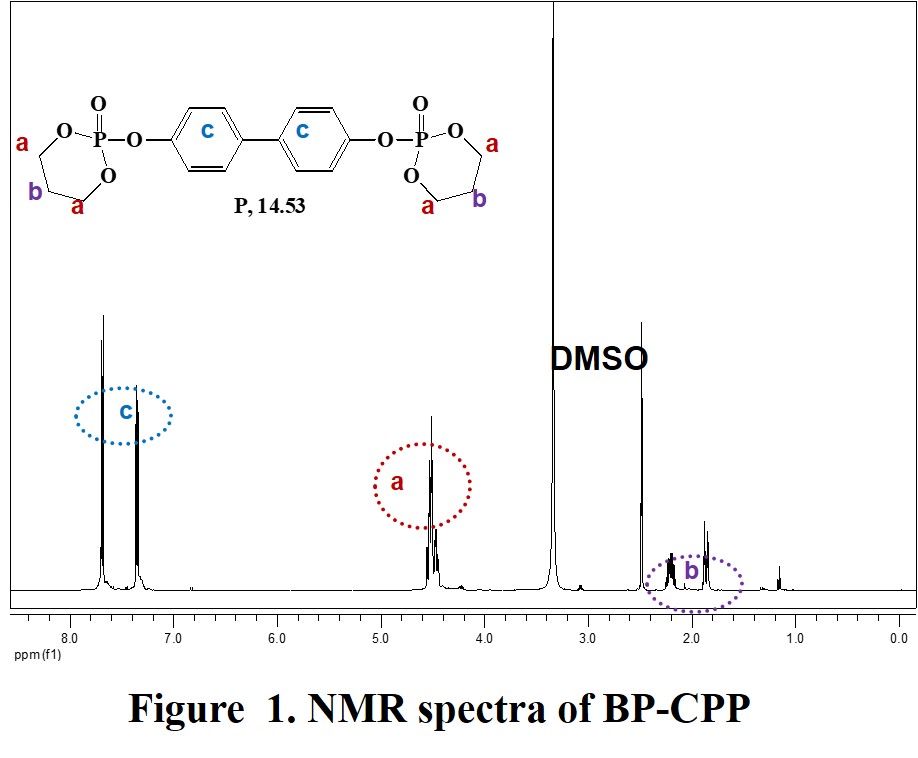
Porous materials have been widely applied to the different fields, such as adsorbents, heterogeneous catalysts, drug loading and delivery. In the current studies, the porous materials are mostly constructed by gas foaming, photolithography, templating, etc. Here, we report an innovative manufacturing process for the fabrication of three-dimension porous structures containing other ingredients including the organic and inorganic via the chemical vapor deposition (CVD). The porous materials were made by poly-para-xylenes (PPXs) replacing the origin sublimating templates via the sublimating/deposition process, and the ingredient that cannot be sublimated would be encapsulated within the PPXs porous materials. The mechanism of the porous structures is that the pores are formed by the gas vapor occupying the remaining vacancies lifted by sublimated template. By adjusting the sublimation rate of the template and the rate of PPXs deposition, the porosity can be controlled. In this report, we present three examples including the magnetic porous particles, cell-containing scaffolds and the protein-encapsulated porous particles. This novel approach overturns the general concept that vapor deposition forms uniform thin film and also provides another way to manufacture multifunctional porous materials.
In this study, a oriented organometallic frame/polyvinyl alcohol (MOF/PVA) nanofiber material was prepared by electrospinning. PVA was uniformly mixed with a MOF to form a solution. In this experiment, zinc nitrate Zn(NO3)2) and 2-methylimidazole were used to synthesize ZIF-8 powder material by in-situ process, and then added to 40mg, 120mg and 200mg respectively. The 10wt% PVA polymer solution was thoroughly stirred, the precursor was disposed, and the electrospinning process was performed. The crystal structure and scanning electron microscope (SEM) were detected by X-ray Diffaction (XRD). Observing the trend of fiber arrangement and the surface morphology of ZIF-8 grown on the fiber, the weight change of the sample detected by Thermogravimetric Analysis (TGA), and the Fourier Transform Infrared Spectroscopy (FTIR) test sample The change of properties before and after mixing of different contents.
As a results, it was found by SEM observation that PVA/ZIF-8 10 wt% was formed by bead-like fibers after electrospinning. From this phenomenon, ZIF-8 was uniformly dispersed in a polymer solution to reunite ZIF-8 by electrospinning technology. ZIF-8 can be evenly distributed in the fiber. XRD analysis showed that there were indeed various characteristic peaks of ZIF-8 in the composite fiber, and it was further confirmed that ZIF-8 was contained in the fiber.
Metal-organic frameworks (MOFs) are porous material composed of metal ions and organic ligands, which have ordered crystalline structure with micro pores and high surface area. MOFs are recently focused as materials for various applications. In the development of the MOF-based mixed-matrix membranes for membrane separation processes, the size and the shape of particles are the important factor for membrane performances. Zeolitic imidazolate framework-L (ZIF-L) is one of the two-dimensional MOF which is focused as materials for mixed matrix membrane; however, the control of the shape and the particle size has not been sufficiently reported yet.
In this study, we report the effect of surfactants on the synthesis of ZIF-L. Surfactants can adsorb on the specific surface of the MOF crystal in synthesis, and can control the size and the shape of the resultant MOF particles. In the synthesis of ZIF-L, we used cetyltrimethylammonium bromide (CTAB) as cationic surfactant, sodium dodecyl sulfate (SDS) and sodium dodecylbenzene sulfonate (SDBS) as anionic surfactants, TWEEN(R) 20, 40, 80 as nonionic surfactants. We synthesized ZIF-L in the presence of various surfactants with various concentrations and characterized the crystal structures using X-ray powder diffraction, the size and the shape of the particles using scanning electron microscope, porous structure using nitrogen adsorption measurements. The characteristics of obtained ZIF-L particles are highly influenced by the species of surfactants, and the details will be reported in the presentation.
Metal-organic frameworks (MOFs) are novel porous materials composed of metal ions and organic ligands. MOFs have ordered porous structures and show adsorption and molecular sieve properties. As one of the applications, MOF-based mixed-matrix membranes (MMMs) have been reported for gas separation. The morphology of MOF particles is the important factor for membrane performances. It is reported previously that MMMs prepared by stacking two-dimensional (2D) MOFs horizontally in the membrane plane can exhibit high gas selectivity by reducing the permeation of non-permeable component. Zeolitic imidazolate framework-7-III (ZIF-7-III) is one of the 2D MOFs which is composed of zinc ion and benzimidazole and forms layered crystal structure with micropores which diameter is approximately 0.29 nm. In the previous report, composite membrane was prepared using ZIF-7-III nanosheets which was made by exfoliation of layered ZIF-7-III particles. The initial ZIF-7-III particle size should be influential on membrane performances; however, there has been no report on the control of ZIF-7-III particle size.
In this study, we report the control of ZIF-7-III particle size using water-in-oil microemulsion. Microemulsion was prepared using heptane as continuous phase, water as dispersed phase, cetyltrimethylammonium bromide (CTAB) as surfactant and 1-hexanol as co-surfactant. Synthesis of ZIF-7-III was conducted in microemulsion, and obtained white powder was characterized using powder X-ray diffraction and scanning electron microscope. Reaction conditions such as reaction temperature, concentration of CTAB were highly influential on the morphology of resultant ZIF-7-III particles. The details will be reported in the presentation.
Metal-organic frameworks (MOFs) are an emerging class of nanoporous materials. Owing to their highly ordered nanopores, MOF membranes are promising for higher selectivity and permeability than polymer membranes which dominate industrial membrane separation. However, high cost of support materials and lack of robust membrane fabrication methods are problematic. Here, to establish a versatile fabrication method, we investigated the formation process of MOF membranes on hollow fibers, originally used for water purifier filter, which are cheap and easily available.
Here we focused on zeolitic imidazolate framework-67 (ZIF-67) composed of Co2+ ions and 2-methylimidazole (MIM) linker, which is a family of MOFs, and applied a secondary growth method. In this method, seed crystals were deposited on the fiber by immersing the fiber in MIM and Co(NO3)2 aqueous solutions alternately, followed by the seed crystals growth by immersing seeded fibers in a mixed aqueous solution of MIM and Co(NO3)2 (growth solution). We first investigated effects of concentration ratio of growth solution (R = [MIM]growth / [Co2+]growth). A small R resulted in the deposition composed of needle-like crystals with many gaps (Fig. 1a), while a large R produced a thin film densely packed by rhombic dodecahedral crystals (Fig. 1b). However, resultant films showed no separation selectivity.
To improve the membrane performance, we modified the growth process. Immersing seeded fibers in a growth solution with a small R was followed by subsequent growth under a large R so that ZIF crystals first grow vertically and then merged with each other by filling gaps in the second growth. This two-step process successfully produced a thick and dense membrane (Fig.1c) with separation factor of 1.15 for air and propane, which suggests that the two-step growth reduces cracks. Hence, we demonstrated that the stepwise control in the crystal growth direction enables the membrane densification.

Metal-organic frameworks (MOFs) are new porous materials discovered in the 20th century. MOFs are synthesized through self-assembly of metal ions and organic ligands, and they have regular arrays of micropores and a large surface area. In addition, by selecting components, MOFs exhibit various crystal structures with tunable pore size. From these characteristics, MOFs are expected to be applied to the medical field, especially drug delivery systems (DDSs). In DDSs, the drug with necessary amount are transported to the right place at the right time by spatial and temporal control. Generally, a carrier for containing or binding the drug is required for DDSs. Although it is common to use a liposome and a polymeric micelle as the carrier, the high inclusion method of drugs has not been established. In recent studies, therefore , MOFs have attracted much attention as a new DDS carrier with high drug inclusion.
In this study, the drug-release behavior from MOFs by a pH change was investigated. As model MOFs, we selected zeolite imidazolate framework-8 (ZIF-8) and Universitetet i Osolo-66-NH2 (UiO-66-NH2). It is reported that the crystal structures of ZIF-8 and UiO-66-NH2 collapse under acid and base conditions, respectively. As model drugs, indomethacin and ibuprofen were selected. We found that ZIF-8 included indomethacin and its dissolution rate from ZIF-8 was the fastest at pH 5.0. On the other hand, UiO-66-NH2 included ibuprofen and its dissolution rate from UiO-66-NH2 was the best at pH 8.5. These results will lead to the development of the new pH responsive DDS with high drug capacity.
Hydrogels are used in applications such as separation, reaction, and drug delivery systems. The diffusivity of solute in hydrogels is an important parameter. For example, the concentration and the diffusivity of solutes, such as oxygen and NH4+, in the hydrogel can affect the activity of ammonia-oxidizing bacteria (AOB) immobilized within the hydrogel for an ammonia wastewater treatment system. The goal of this study is to develop a novel method to measure the diffusivity of solute in spherical hydrogel using a microsensor. A microsensor measures the chemical environments at the scale of the micrometer-sized sensor tip, as well as the concentration of solute in hydrogel. Millimeter-sized spherical hydrogel is suitable for the experiment using the microsensor and the analysis of diffusion kinetics. The spherical polymeric hydrogel was prepared using a production method combining sedimentation polymerization and two-fluid atomization, developed in our previous study. Oxygen, NH4+, and N2O were used as model solute for AOB-catalyzed reactions. The experiment on oxygen diffusion was carried out as follows. Firstly, a microsensor was inserted in the center of the spherical hydrogel and the hydrogel was placed in anaerobic water purged with N2 gas. Then, water was purged with air and the dissolved oxygen (DO) concentration in the hydrogel was measured continuously. The DO concentration was approximately 0 during initial period, and increased with time up to approximately 230 mmol/m3 in air-saturated water. Diffusivity of solute in hydrogels was successfully analyzed using the solution of the Fickian diffusion equation within a sphere.
Pd catalyzes various kinds of reactions such as automobile exhaust emission treatment and organic synthesis. In this study, we focus on polymer gel as a heterogeneous catalyst carrier. The gel is a polymer of a three-dimensional network structure swollen with a solvent and can be handled like a solid, but the inside of the gel is also like a liquid. The aim of this study is to develop a polymer gel supported Pd catalyst using various polymer gels, to clarify the influence of the absorption and adsorption ability of the gel and the holding state of Pd(II) ion on the catalytic activity to construct a design guideline for a high performance gel catalyst. The model polymer gel was copolymer gel of allyl mercaptan (AM) having a thiol group interacting with Pd(II) ion and multicomponent, and gel having amino group and ammonium group in side chain. The various kinds of gels were synthesized by free radical polymerization. Pd(II) ion-adsorbed gels were prepared through the Pd(II) ion adsorption experiment, and subsequently Pd nanoparticle-loaded gels were prepared by the reduction of Pd(II) ions using NaBH4. The model organic synthesis was Suzuki-Miyaura coupling (synthesis of biphenyl with phenylboronic acid and iodobenzene as reactants) using both catalysts Pd(II) ion-adsorbed gels and Pd nanoparticle-loaded gels. The adsorption properties of Pd(II) ions of various gels, characteristic properties of gel supported Pd, and Suzuki-Miyaura coupling reaction characteristics using gel supported Pd were investigated.
The excited electrons and holes generated upon by irradiating UV ray to TiO2 have powerful redox power. They produce reactive oxygen species (ROS) under aerobic conditions at the surface. These species rapidly decompose organic compounds to convert them into CO2 and H2O via carbon-carbon bond cleavage. On the other hand, they can also catalyze organic synthesis reactions such as carbon-carbon bond formation. The reactions must be carried out in dry anaerobic conditions in order not to produce ROS that can damage organic compounds. Usually, TiO2 has been used as a powder to increase the catalytic effect. However, the use of particles involves the need of incorporating extra stages in the purification process, such as separation and catalyst recycling. We focused on a composite of TiO2 nanoparticles and a polymer gel in which TiO2 nanoparticles are dispersed and immobilized in the polymer gel as a heterogeneous catalyst. The purpose of this study is to develop a novel TiO2 nanoparticle-loaded polymer gel and clarify its applicability as a catalyst for organic synthesis reaction. As polymer gels, N,N-diethylacrylamide gel, N,N-dimethylacrylamide gel, and N-isopropylacrylamide gel were used. Commercial TiO2 nanoparticle (AEROXIDE® TiO2 P-25) with 21 nm in diameter was used. The composite gels were synthesized by free radical polymerization. The 4-iodetoluene dehalogenation reaction as a model organic synthesis reaction was performed by a batch operation using 4-iodetoluene, diisopropylethylamine, dried composite gel, and acetonitrile as a solvent. The reaction was irradiated with UV light and stirred at room temperature under a nitrogen atmosphere. The composite gels catalyzed the reaction successfully.
Sodium alginate aq. is known to form hydrogel on mixing with CaCl2 aq., where Ca2+ works as the crosslinker. Megascopically, the above process is observed as instantaneous gelation. The authors have been interested in capturing the quick gelation in terms of how fast it occurs on actual mixing of these two starting aqueous solutions. First, 1wt% sodium alginate aq. was added dropwise into 10M CaCl2 aq. and the time evolution of the morphology of the droplet after the impingement onto the surface until the stabilization of its outer surface was recorded in a high-speed motion picture. The difficulty in fulfilling our observational purpose relying on the above method was revealed that the gradual morphological alteration toward the spherical geometry is temporarily too moderate to be detected by the high-speed motion picture method. In order to capture the arrest in the deformation of the gelling solution more effectively, we needed to choose a situation where the tiny surficial undulation of the gelling solution is incessantly induced by being in contact with 10M CaCl2 aq. In the intensively discharged gelling solution out of an injection syringe, it was found that the straight linearity of the gelling solution in the translational motion was observed while it kept in the state of a flowable solution. The beginning of the deviation from the as-injected linear flow implied the vanishing fluidity. Thereafter, the gelling solution was seen to behave like rod-like objects forming an entangled coil. From the discharging velocity of the gelling sodium alginate aq. at the exit of the injection syringe, the timescale for the gelation was estimated to be 10-3s in the order of magnitude.
In many countries, arsenic level in groundwater exceeds the acceptable limit. Although we removed Arsenate in our previous research, removal of Arsenite is harder. The purpose of the study is to adsorb Arsenite, As(III), from ground water by adsorption using a composite of cationic polymer gel impregnated with iron hydroxide. The cationic polymer gel that we used is N,N-dimethylamino propylacrylamide, methyl chloride quaternary (DMAPAAQ).
The gel was prepared in a unique method where Sodium Hydroxide and Iron (III) Chloride was added to the monomer and initiator solutions, respectively, in order to maximize the FeOOH contents in the polymer structure of the gel.
Transmission Electron Microscope (TEM) images show that our gel contains visible FeOOH particles in its structure. The Thermogravimetric Analysis (TG) indicates that the maximum weight percentage of FeOOH particles in the gel is 53.7%. Generally, there are three types of FeOOH. We performed Mössbauer spectroscopy to determine the type of FeOOH contained in the gel. The results match with that of γ-FeOOH. Previous arsenic removal experiments found that γ-FeOOH helps to remove both As(III) and As(V). Our studies suggest that in the structure of DMAPAAQ + FeOOH gel, the polymer structure of DMAPAAQ adsorbs 35.6%, whereas γ-FeOOH components adsorb 64.4% of arsenic. Hence, DMAPAAQ + FeOOH can adsorb arsenic successfully. FTIR analysis suggests that when As(III) and As(V) were adsorbed by the DMAPAAQ + FeOOH gel, changes such as shifts, appearances and disappearances in several surface functional groups occurred. These results further prove the adsorption of arsenite to the gel.
Our study successfully showed that the newly developed gel composite, DMAPAAQ and FeOOH, adsorbed As(III) effectively because of having γ-FeOOH components in the gel structure. The novelty in our research is in the unique preparation method of the gel, and It's characterization.
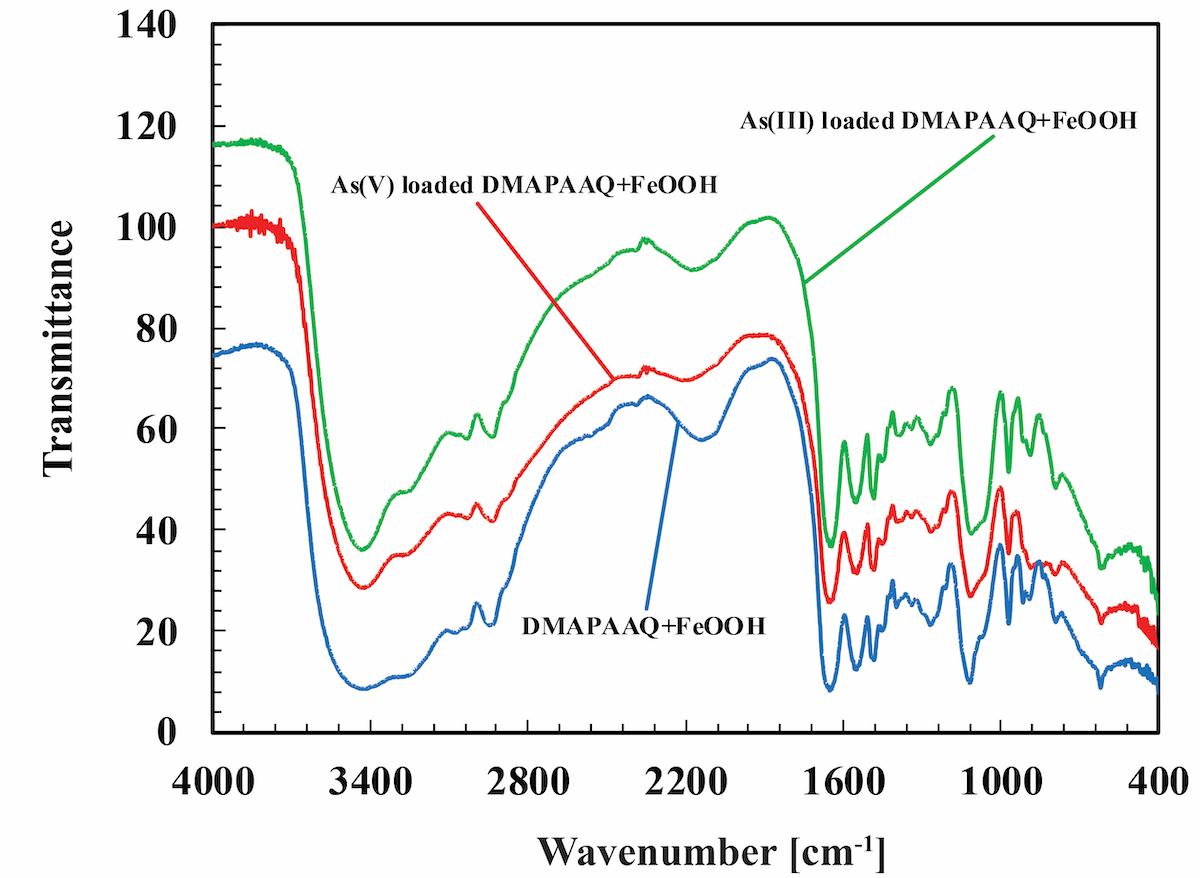
Catalysis by Heteropoly acids (HPAs) and polyoxometalates (POMs) having a higher demand worldwide, as it can be designed to accelerate complex reactions and be more environmentally friendly. However, recycling of water-soluble solid catalysts remains a problem. The synthesis of a recyclable composite with catalytic properties is the key to better use of HPAs and POMs. Many researches have mentioned the method of synthesis by immersing a porous carrier in a supported solution. However, the catalytic abilities of the previously studied composites after multiple uses have rarely been mentioned. In this research, a novel idea is proposed to synthesize a heteropoly acid supported composite. A complex hydrogel with catalytic properties was synthesized by mixing an anionic monomer with a heteropoly acid. The heteropoly acid particles were inserted inside the hydrogel by the interaction forces between the anions. Thus, preventing the water-soluble heteropoly acid from being lost during the catalytic reaction. The complex hydrogel is consisted of the anionic monomer 2-acrylamide-2-methylpropanesulfonic acid (AMPS) as a carrier, N,N′-Methylenebisacrylamide (MBAA) as a crosslinker and the typical keggin-type HPA: H3PW12O40, which was synthesized by one-pot method. Then the composite is divided into five groups. Following this, the five groups were washed by deionized water for 24h, 48h, 72h, 96h and 120h respectively. The existence of H3PW12O40 crystalline nanoparticles in the composite was confirmed by X窶甚ay diffraction (XRD). After comparing the samples from different groups by Scanning Electron Microscope (SEM), all the samples have similar surface structure and nanoparticle aggregation state. Nanoparticles were observed from the samples of each group by Transmission Electron Microscope (TEM). From the thermogravimetric analysis (TGA), the particle content rate of samples from each group was kept at about 17% as the washing time increased. It indicates a higher potential of this synthetic method to a wide range of applications.
Removal of heavy metals from ground water and soil has been a common problem both in industry and agriculture, since high concentration of heavy metals are harmful to environment and human health through water and food. In this study, a simple and effective removal method of various harmful anionic ions such as arsenic acid, selenium acid, and chromium acid from soil and water was proposed. Generally, these anions were removed by cationic adsorbent or cationic ion exchange resign. However, various adsorbents have low ability to remove heavy metals from low concentration solutions, and it sometimes desorb those ions when interfering ion exist. We present new ion removal method by using hydrogel. Anionic hydrogel of 2-Acrylamido-2-methylpropanesulfonic Acid Calcium salt (AMPS-Ca) and cationic hydrogel of N,N-Dimethylamino Propyl Acrylamide (DMAPAA gels) were used to remove anionic heavy metal ions. Each heavy metal ion could be partially removed by both hydrogels. It was found that removal of arsenic and selenium by AMPS-Ca hydrogel as attained by the formation of heavy metal calcium salts in the hydrogel. On the other hand, the crystallinity of the heavy metal salt was not found in the DMAPAA gel. It is found that removal of heavy metals by each hydrogel was made by adsorption or formation of heavy metal salt in the hydrogel. In addition, the various hydrogels used in this study could absorb water as well as sodium acrylate. Therefore, it is suggested that they also provide a method of solidify the heavy metal contaminated sludge discharged in tunnel excavation work. The contaminated sludge will be recycled after the treatment of the hydrogel.
Introduction
The demand for lithium-ion batteries has rapidly increased in recent years, and the supply will be insufficient in near future. Therefore, the establishment of lithium recycling process is required to solve the problem of the increase of the waste lithium batteries. However, conventional lithium recovery processes are complicated and costly. We have presented new simple lithium recovery process1) using hydrogel with a quaternary amine ((3-Acrylamidopropyl) trimethyl ammonium chloride)(APTAC). In this research, the reaction mechanism of lithium and carbonate ion are investigated.
Result and discussion
A schematic diagram of the process is shown in Figure 1. At first, APTAC gel was immersed in the solution containing anion which exchanged Cl-. The gel was then immersed in LiCl aqueous solution. The Li+ reacted with the anion in the gel to produce lithium salt. The Li+ was recovered as the lithium salt within the gel.
We investigated which anion was suitable for Li+ recovery and found that the HCO3- recovered the lithium ion best among PO43-, HCO3- and CO32-. HCO3- reacted with Li+ more than their stoichiometric ratio, 1.0, when the initial concentration of Li+ was more than 1000 ppm. It is considered the Li+ initially reacted with HCO3- to form metastable LiHCO3 when its initial concentration was below 1000 ppm. Then H+ in a part of the LiHCO3 exchanged with Li+ in the solution to form stable Li2CO3 when it was higher than 1000 ppm. Therefore, Li+ was recovered more than double of the stoichiometric ratio.
The elemental composition of the gel after the Li+ recovery was analyzed by XPS. The gel had the C-O single bond that was not exist in the original APTAC gel. We found that Li+ reacted with HCO3- and was recovered as LiHCO3 or Li2CO3 in the gel.
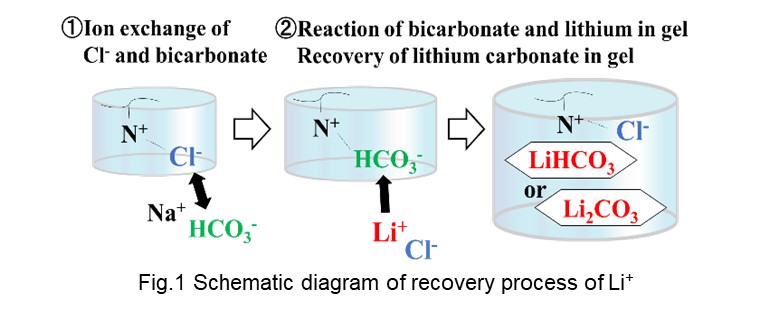
Gel bead can be used as a carrier for drugs and fertilizers and its diffusion characteristics has been studied for controlled release of chemical substances. In our study, the effect of the addition of the dispersed phase on the diffusion rate of substances in hydrogel was investigated. The oxygen diffusion rate in hydrogel containing microbeads was measured to study the effect of the size and amount of microbeads on the diffusion inhibition.
Figure 1 shows the effect of microbead addition on apparent diffusion coefficient of oxygen in hydrogel. The apparent oxygen diffusion coefficient was calculated by solving one-dimensional unsteady diffusion equation with measured data of dissolved oxygen concentration profile in the gel. First, the apparent oxygen diffusion coefficient in the gel containing the microbeads was smaller than that without the microbeads. It was confirmed that the addition of the microbeads inhibited oxygen diffusion.
Next, the apparent diffusion coefficient decreased with an increase in the microbeads holdup. On the other hand, the diffusion rate did not depend on the microbead size. To develop the estimation method of apparent diffusion coefficient, diffusion in hydrogel containing microbeads was considered by analogy with the diffusion theory in porous medias (Ohashi et al., 1993) where diffusion path is lengthened by solid obstacles. The experimental values agreed well with the model. This suggests that the apparent diffusion coefficient can be estimated from the volume fraction of the added dispersed phase.
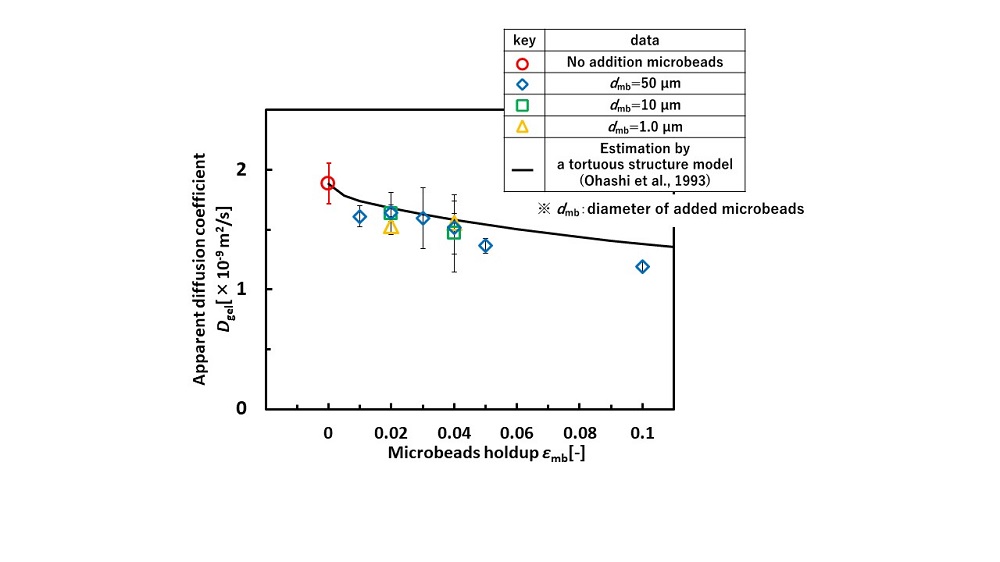
Heat dissipation is one of the most important issue for designing high performance devices. Thermal interface materials (TIMs) are used to promote heat transfer at the solid-solid interface between IC chip and heat radiator in electronic devices. Presently, composite materials of thermally conductive fillers and polymers are widely used, however polymers have problems of low thermal stability and low thermal conductivity. Here, we propose and report Ag aerogel films composed of Ag foils sandwiched with Ag porous layers (Fig. 1a,b). The film was fabricated by simultaneously forming Ag particles by gas-evaporation and depositing Ag particles on an Ag foil. The boat-foil distance was carefully optimized to make the Ag porous layer soft and uniform. The steady-state measurement showed a thermal resistance as small as <20 mm2K/W (Fig. 1c), which is similarly small as the high performance TIM of an indium sheet. Our aerogel TIM, made of Ag having a melting point (962 °C) much higher than indium (157 °C), can expectedly be used for thermal management under high-temperature conditions.

Hydrogel have good flexibility and biocompatibility, so they cause little damage by implantation in the body. Furthermore, since hydrogels can release drugs, they are expected to be applied to a local drug delivery system. The amount of drug used can be reduced and side effects can be alleviated by the local drug delivery system. However, the hydrophilicity of hydrogel interfere result in suppression the controlled release of hydrophobic drugs such as anticancer agents or antibiotics. We have developed hydrophobically-modified gelatin (HMG) and could prepare a hydrogel by only hydrophobic interaction. HMG hydrogel do not require the use of chemical crosslinkers, which are often toxic to the body. In addition, HMG hydrogel have hydrophobic segments in its structure, which can absorb hydrophobic drugs and expect to control their release. As a factor to control the amount of adsorbed hydrophobic drug in the hydrogel, we focused on the strength of hydrophobicity of hydrophobic group. HMG with different alkyl chain length (C4~C12) of modified hydrophobic group were synthesis. The swelling ratio of these hydrogels were decreased in increasing the chain length of alkyl group modified to HMG. This result means that the increase of alkyl chain length intensifies the hydrophobic interaction. Then, HMG hydrogel was adsorbed with uranine used as a model for hydrophobic drugs. The adsorption test exhibited that the amount of hydrophobic drug adsorbed in HMG hydrogels could be controlled by varying hydrophobic alkyl chain length. The controlled release of the drug was confirmed by drug release experiments in vitro. In conclusions, HMG hydrogel could control drug adsorption and delivery.
Novel organic-inorganic hybrid material was developed as a LED sealant.
Polysilsesquioxane with -SH group (PSQ-SH) and acrylic monomer were selected as inorganic and organic components, respectively. Trimethylolpropane Trimethacrylate (TRIM) which is a trifunctional monomer, has been selected as an acrylic monomer, and curing of the material was performed by the Thiol-ene reaction.The LED light emitting substrate was sealed using the prepared PSQ-SH/TRIM hybrid material, and a light emission test was performed. However, there was a problem that cracks occurred in the hybrid material during lighting. Therefore, Ditrimethylol propane tetraacrylate (DTMPA), which is a tetrafunctional monomer, was newly selected to prepare a PSQ-SH/DTMPA hybrid material in order to enhance the strength and heat resistance of the material. The organic component can be reduced by the increasing the number of the functional group in the monomer, and a more inorganic rich material can be prepared. The heat resistance can be improved by increasing the inorganic components in the hybrid material. The mechanical strength of the hybrid material can also be improved by increasing the polymerization density. Therefore, the characteristics of novel PSQ-SH/DTMPA hybrid material were evaluated and compared with PSQ-SH/TRIM hybrid material. As the contents of the evaluation, the heat resistance was evaluated by confirming the weight reduction start temperature using the Thermalgravimetry-differential thermal analyzer(TG-DTA) analysis, the mechanical strength was also evaluated by bonding the glass substrates with a hybrid material and measuring the sealing strength. The obtained results did not improve with respect to the heat resistance even if the monomer was changed.However, the mechanical strength was greatly increased by changing the monomer.
In conclusions, it was possible to suppress the generation of cracks at the time of lighting by using the PSQ-SH/DTMPA hybrid material compared to PSQ-SH/TRIM hybrid materials in the light emission test.
Rod-shaped nitroxide radical (NR) compounds with a five-membered ring NR moiety in the mesogen core (LC-NRs) show unique intermolecular magnetic interactions in their liquid crystalline (LC) and isotropic liquid (Iso) phases above room temperature; their magnetic susceptibilities increase abruptly at the phase transitions from the crystalline (Cr) to LC phases. This phenomenon is called magneto-LC effects. This effect is considered to originate from inhomogeneous intermolecular magnetic interactions. In fact, we confirmed that the effect was enhanced by adding a small amount of (2S,5S)-2 to (2S,5S)-1 (Figure). We also synthesized a new LC-NR with an azobenzene moiety (2S,5S)-3 whose trans and cis isomers have similar shapes to (2S,5S)-1 and (2S,5S)-2, respectively. Since an azobenzene moiety shows the photoisomerization, we expected to change reversibly in inhomogeneity of intermolecular magnetic interactions of (2S,5S)-3 due to the change of the molecular shapes during the photoisomerization. Here, we report the synthesis of a new LC-NR with an azobenzene moiety in the mesogen core and the effect of the photoinduced phase transitions on magnetic properties.
We observed the phase transition behaviors under ultraviolet (UV) or visible (Vis) light irradiation by polarized optical microscopy and measured UV-Vis light absorption spectra. We also measured electron paramagnetic resonance (EPR) spectra under continuous irradiation of UV or Vis light.
Photoinduced phase transitions occur only between LC and Iso phases under UV or Vis light irradiation for (2S,5S)-3. This is likely to arise from the increase of the ratio of cis-isomer by the UV light irradiations, which could reduce the orientational order parameter. EPR spectra indicate that magnetic susceptibility increases under UV light irradiation and decreases under Vis light irradiation. We confirmed that bidirectional switching of the magnetic properties of (2S,5S)-3 occurs under alternating irradiations of UV and Vis light.
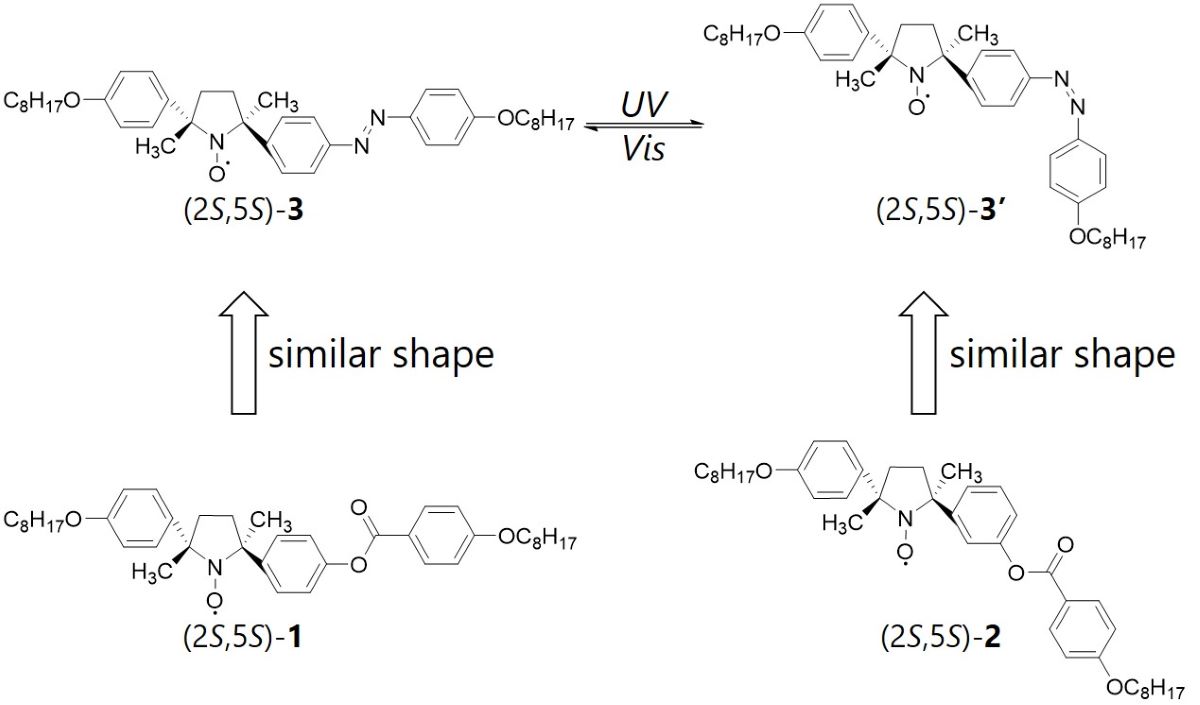
With the increasing demands on wearable devices, the stretchablity of various electrical devices have drawn much attention recently. The most commonly used method to enhance mechanical performance of stretchable polymers is introducing nanofillers to form a composite system. Polydimethylsiloxane (PDMS) is the most widely used silicon-based elastomer, and has been widely used in many medical, biological, and microfluidic applications. In the literature, various nanofillers, such as carbon nanotubes, graphene sheets, and silver nanowires, have been well dispersed in PDMS to demonstrate the flexibility and versatility of PDMS composites.
On the other hand, because of the chemical inertness, it is difficult to include ionic fillers in PDMS. PDMS composites with ionic filler usually have problems like filler aggregation, delamination, phase separation, or fractures, under stretching conditions. To address this problem, in this work, a water-in-oil type emulsion is used as a carrier for water-based ionic materials. The relationship between applied sheer rates and droplet size will be examined to elucidate the emulsion process. Moreover, emulsion stability will also be carefully characterized to investigate the effectiveness of various emulsifying agents. In summary, this research provides a new method for PDMS composite synthesis and opens a new avenue to stretchable polymer applications.
In the last two decades, many reports revealed the novel functional properties of natural and synthetic peptide lipids. Previously, we also reported the high self-assembling ability of various peptide lipids and demonstrated high cytotoxicity of some peptide lipids to animal cells. Peptides can be substrates for many enzymes in a living system. For example, tyrosine kinase adds a phosphate group to a tyrosine residue in a target protein, which plays a key role in signal transduction on cell growth and differentiation. Gene mutation in tyrosine kinase leads to uncontrollable signal transduction and causes many cancers. In other words, tyrosine kinase activity of some cancer cells is different from other cells.
Here, we prepared novel peptide lipids that underwent phosphorylation of its peptides by tyrosine kinase in animal cells and evaluated their cytotoxicity. First, we synthesized different types of peptide lipids by solid-phase peptide synthesis, which were composed of fatty acids (C8 – C16) and peptides containing tyrosine (Fig. 1). The peptide sequences were designed according to tyrosine kinase substrates. The synthesized peptide lipids were purified by HPLC and confirmed by MALDI-TOF MS. When a phosphate group is added to tyrosine in a peptide lipid by tyrosine kinase, the hydrophilicity of the peptide lipid increases. We conjectured that a peptide lipid is transformed to anti-cancer drugs in cancer cells. We expected that phosphorylated peptide lipids should have cytotoxicity different from the original peptide lipids (Fig. 1). The present study proposes the novel effective treatment of drug-resistant cancer cells based on kinase activity.
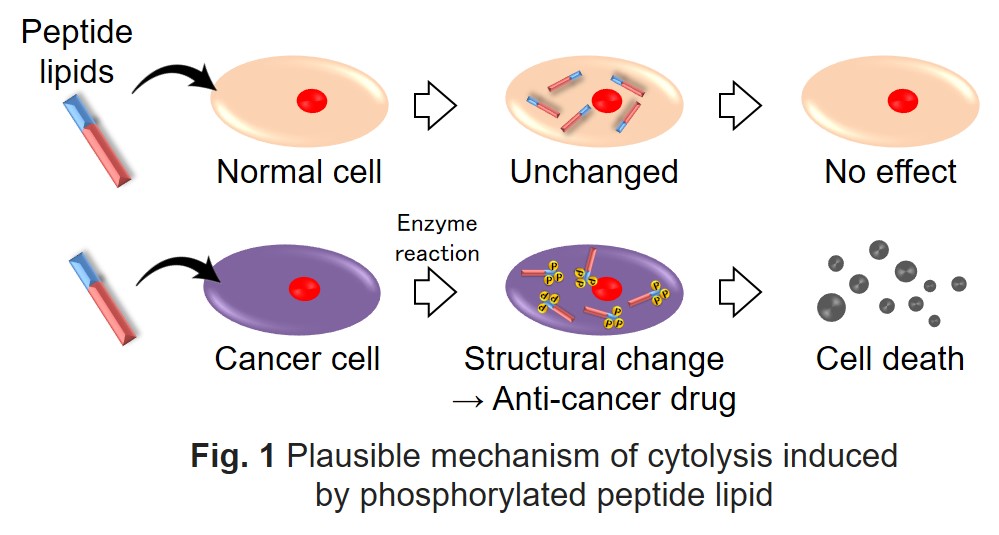
Lipid vesicles are closed bilayer membrane(s) consisting of amphiphilic lipid molecules such as phospholipids formed in aqueous media. Lipid vesicles can encapsulate various water-soluble biochemical molecules into their internal water phase, therefore lipid vesicles have been studied as biochemical microreactors surrounded by lipid bilayer membranes, those are structurally similar to biological cells. However, the utilization of lipid vesicles as microreactors is limited by their preparation method because it is generally difficult to obtain vesicles containing biochemical molecules such as enzymes with high encapsulation efficiency and controlled diameter.
Recently, we developed a novel vesicle preparation method by the combination of multiple emulsification and solvent evaporation techniques (Kuroiwa et al.: J. Am. Oil Chem. Soc., 93, 421-430, 2016). In this method, at first, water-in-oil (W/O) emulsion containing water-soluble enzymes was prepared by membrane emulsification. Next, water-in-oil-in-water (W/O/W) multiple emulsion containing above W/O emulsion as a dispersion phase was formulated by microchannel emulsification with water-soluble polymeric emulsifiers. After removal of organic solvent in the oil phase of W/O/W emulsion by solvent evaporation under the ambient condition, finally we obtained enzyme-containing lipid vesicles. The diameter of lipid vesicles reflected that of the starting W/O emulsion droplets. For porcine pancreas lipase, the activity-based encapsulation yield was determined to be ca. 30%. These value was quite high compared with values for conventional vesicle preparation methods. Lipase-catalyzed reaction inside lipid vesicles was carried out by supplying a substrate (fluorescein diacetate) from the external water phase to internal water phase of lipid vesicles. The hydrolysis of the substrate was proceeded as comfirmed by fluorescence of hydrolytic product, fluorescein, from internal water phase of vesicles. This result suggests that the substrate permeated through lipid bilayers of vesicles and then it was hydrolyzed by lipase inside vesicles. We also evaluate the encapsulation characteristics of a proteolytic enzyme.
Structural color is color produced from materials having microscopic structures that can interact to visible lights via interference and/or scattering phenomena. Artificial structured materials have been widely developed by fabricating various nano-sized structures using synthetic organic or inorganic materials. In contrast, development of structural colored materials based on natural biomolecules have not studied enough although very vivid structural colors are widely observed in nature, for example, in butterfly wings, beetle shells, and bird feathers.
In this study, we developed a novel structural colored material using millimeter-sized molecular aggregates consisting of natural amphiphilic lipids including phospholipids. A lipid mixture consisting of phosphatidylcholine, cholesterol, and additives (various monoalkyl compounds) was dissolved in n-hexane and placed onto aqueous phases with various pH values. After evaporative removal of n-hexane, molecular aggregates with visible size (larger than 1 mm) were formed in the aqueous phase. The shape of molecular aggregates was film-like or spherical, depending on lipid composition and pH value in the aqueous phase. Small/wide angle X-ray scattering measurements revealed that the molecular aggregates exhibited the liquid crystalline polymorphism and their phase structure was related to their macroscopic shape. Among them, spherical aggregates with cubic liquid crystalline phase could be converted to aggregates exhibiting colorization by controlling the salt concentration of their external aqueous phase. The reflection spectra of visible lights from the colored molecular aggregates suggested that their colorization was attributed to the internal structure of the molecular aggregates. Electron microscopic analysis demonstrated the existence of periodic structures with the characteristic distance corresponding to wavelength of visible lights. We believe that our finding would contribute to development of novel lipid-based colorization materials that are potentially applicable for foods, cosmetics, and display materials.
Amphiphilic molecules form self-assembled aggregates, such as micelles and vesicles. These aggregates are expected to be applied as a drug carrier and a microreactor. Polarity of the hydrophobic region is an important parameter to enbed hydrophobic drugs and substances. In this study, the relative permittivity was evaluated by using fluorescent probes. These probes were synthesized by conjugation of 1-pyrenemethanol and dicarboxylic acids, succinic acid (P-C3-COOH), suberic acid (P-C7-COOH) and dodecanedioic acid (P-C11-COOH). Aggregates were prepared by non-ionic sorbitan surfactants, Span 40 and Tween 40. The mixture of these surfactants forms various kinds of aggregates, Tween 40-rich and Span 40-rich mixtures tend to form micelles and vesicles, respectively. Figure 1 shows the relative permittivity of aggregates using P-C3-COOH. Relative permittivity of vesicles at low temperature was apparently higher than that of vesicles at high temperature, it is due to the localization of P-C3-COOH. Rigid structure of vesicles prevented P-C3-COOH to insert into the deep hydrophobic region. Increase of temperature change vesicles more fluid, resulting in decrease of relative permittivity. The measurement using P-C7-COOH shows a similar result as that using P-C3-COOH, while it seems that behavior of P-C11-COOH was different from that of P-C3-COOH and P-C7-COOH. The relative permittivity of micelles did not change regardless temperature. On the other hand, relative permittivity of vesicles was increased from 10 to 30 °C. It was related to phase transition temperature of vesicles. These results help to assume localization of drugs or substances in the aggregates, such as the considering about to apply as a drug carrier or a microreactor.
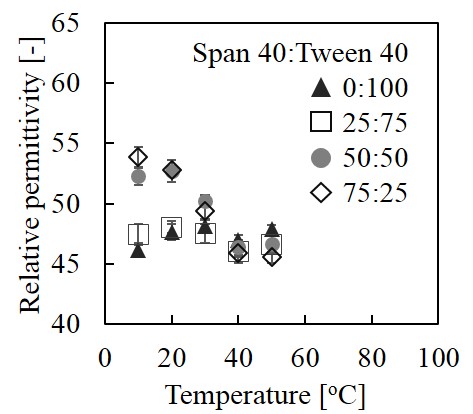
Bicelle is a disk-shaped self-assembly with bilayer membranes like vesicle, and its size is small as well as micelle. In addition, bicelle with a large disc diameter aligns to the magnetic field. Because of these unique properties, bicelle has been attracting tremendous attention as NMR measurement medium. In the formation and application of bicelle, the ordered structure of bicelle bilayer membranes is particularly important. In this study, bicelles were prepared by DMPC and DHPC, then the ordered properties of the bicelle bilayer membranes were characterized by combining the fluorescent probe method and induced circular dichroism spectroscopy (ICD). ICD is a phenomenon that CD activity occurs when achiral molecule DPH is ordered highly within a chirality lipid membrane. The ICD intensities at 360 nm decreased with increasing temperature, suggesting that the motion of the DMPC molecule was limited at low temperature, and the orderly orientation of DPH in the bilayer membrane was lost. The ICD intensity of bicelle was stronger than that of conventional vesicle. Furthermore, the aggregated bicelles, such as stacked bicelle, can be designed by modifying the membrane surface properties of bilayer region.
References: P. Walde et al., Langmuir, 13, 1668 (1997). S. Taguchi et al., Colloids and Interfaces, 2 (4), 73 (15 pages) (2018).
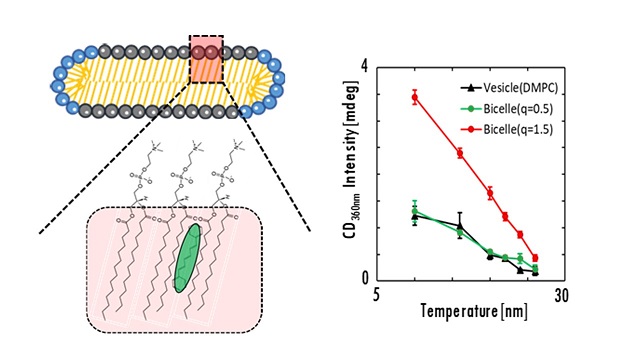
The "gas-liquid two-phase mixed flow" mechanism that fine bubble generation uses rapidly swirling water which shear gas and making small bubbles in water. Based on this mechanism, sending liquid such as oil into the swirling water instead of gas and it able to be emulsified. At present, evaluation methods for emulsions can be considered as particle size distribution and dispersion stability. However, in systems where the separation behavior is not good, it is a problem to catch only some dispersed oil particles and use them as a whole evaluation. In order to express numerical evaluation including the oil particles which can not be dispersed, we use chemical oxygen demand (COD) as numerical evaluation. The figure shows the temporal change of the amount of volatilizing oil calculated from the COD of emulsions sample (1)-(3). The dispersed phase used was of three types: sample (1) only one oil, sample (2) an anionic and a nonionic emulsifier were mixed and added to the oil, sample (3) an anionic and an another kind of the nonionic emulsifier were mixed and added to the oil. It was confirmed that COD decreased and sample (1) has a large amount of volatilized oil because there were many oil particles that can not be dispersed. The performance of the emulsion could be expressed numerically, including the oil particles which could not be dispersed. We also directly analyzed volatile components and compared them with COD results.
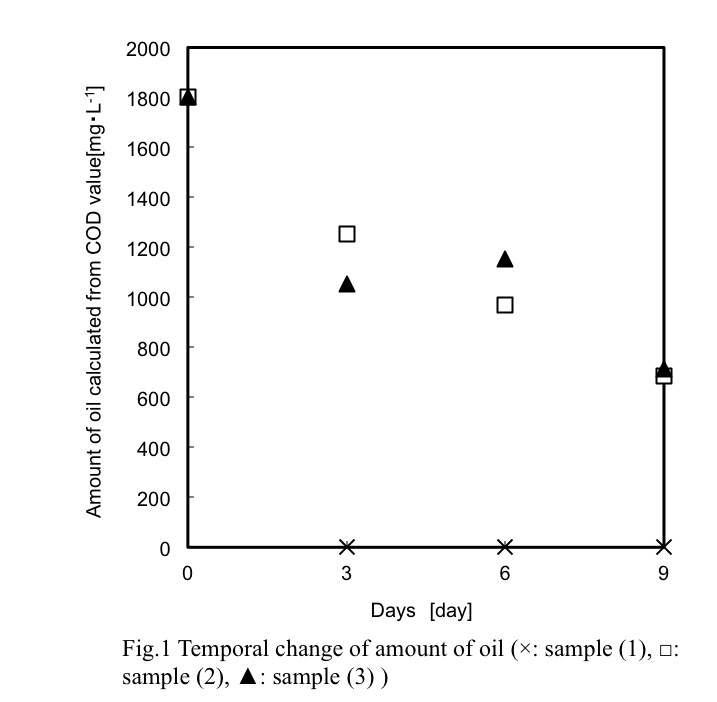
In this study, a plant-derived alkyl polyglucoside (APG) surfactant with alkyl chain of C8- C10 was formulated with the essential oil of Melaleuca alternifolia (tea tree) in the presence of propylene glycol as co-surfactant. This nonionic surfactant with a tradename as Triton CG 110 is reported to possess a CMC at 1748 ppm at 25oC. Typically, tea tree oil (TTO), commonly used in variety of cosmetic and health care products, was extracted from tea tree leaves by Triton CG110-assisted hydrodistillation. The extraction yield was 6.68% under the conditions with (1) 645.4 ppm Triton CG110, (2) at a liquid/solid ratio of 24.5, and (3) for 128 mins as the extraction time. The pseudo-ternary phase diagrams were investigated at the different weight ratios of surfactant mixtures (Smix = CG/PPG) as 1:1, 3:1, 6:1 with hydrodistilled TTO and commercial TTO by water titration method at room temperature. One-phase region consisting of micelles and microemulsion (μ_phi) was painted in gray in Figure 1. The greater the amount of Triton CG110, the wider phase compositions of microemulsion formed. For example, the largest microemulsion region was occurred at a surfactant mixture of 6:1. No liquid crystalline phase was observed at all ratios of Smix under the cross-polarized microscope. Particularly, the effect of storage temperature on selected microemulsion made of 1% TTO with various concentrations of Triton CG100/PPG (3:1 w/w) were recorded. Shelf stability was examined on microemulsions right after preparation and in one month with dynamic light scattering. These results also showed that microemulsion made of 1%TTO, 9% Triton CG100/PPG (3:1 w/w) was insensitive with time and temperature during long time. Antimicrobial activity and antioxidant activity were evaluated with microemulsion made of 1% TTO.
Recently, Pluronic surfactants (PEO–PPO–PEO triblock copolymers, where PEO: poly(ethylene oxide)x and PPO: poly(propylene oxide)y) were found to show CO2-responsive aggregation behavior in water. In earlier paper, mesoporous silica prepared in a hexagonal liquid crystal/water system was swollen by CO2 and the mesopore size was tuned by pressure of CO2 atmosphere. It suggests various potential applications of CO2-responsive surfactant. To clarify mechanism of CO2-responsive behavior of Pluronic surfactant assemblies in water, this study examined effects of temperature, pressure, PEO and PPO lengths on growth of Pluronic surfactant micelles in water under high-pressure CO2 atmosphere.
Aqueous micelles of Pluronic surfactants F108 (x = 150, y = 55) and F127 (x = 100, y = 65) showed no significant change in hydrodynamic diameter of micelles even at high CO2 pressures of 100–300 bar. In contrast with those Pluronic surfactants, F68 (x = 80, y = 30) and F88 (x = 100, y = 20) displayed clear micellar growth with increasing CO2 pressure. Figure 1 shows relationship between the diameter of aqueous micelles and density of CO2 atmosphere at 75 °C and the surfactant concentration 1 wt% for F68, a nonionic EMULGEN 4085 having the C14H29-tail and the PEO (x = 85) close to that of F68, and a nonionic BrijS100 (x = 100) having the C18H37-tail and the PEO chain (x = ~100) similar to that of F88. The tendency that the higher the CO2 pressure (or CO2 density) the larger the micellar diameter was considerably small for nonionic surfactants without a hydrophobic PPO chain, compared with Pluronic surfactants having corresponding PEO units. Especially the Pluronic surfactants with PEO lengths x = 80–85 were found to exhibit significant micellar growth with increasing CO2 density.
Real-time monitoring of small molecules, such as reactants, dissolved gas molecules or metabolic products, during biological conversion is of paramount importance for understanding the metabolic pathway, optimizing conversion efficiency and enhancing the reproducibility of bioprocesses. Generally, detection of such small molecules during biological conversion are largely dependent on chromatography-based techniques, such as high performance liquid chromatography and gas chromatography. However, these techniques are time-consuming, requiring more than 10 minutes for the separation of sample in the column. Moreover, off-line sampling process is inevitable for the analysis which has a risk of causing contamination. For these reasons, development of a novel method that is suitable for real-time monitoring of biological conversion is required. Here, we propose the plasmonic detection of small molecules in biological conversion by using plasmonic nanostructures produced using capillarity-mediated inverse transfer. Nanoparticles assembled at air/water interface are transferred onto solid substrates through the reconstruction of the assembly interface along the direction of gravity by a capillary tube, enabling the transfer of nanoparticles on solid substrate via direct contact. Small molecules are preconcentrated on plasmonic nanostructure through electrostatic attraction and chemisorption. Preconcentration of small molecules increases the number of molecules affected by strong near-field enhancement at the proximity of the metallic surfaces; thereby, allowing the detection of trace amount of small molecules in the solution.
A living matter moves as an efficient energy conversion system under a non-equilibrium state. The study of this motion may be associated with creation of innovative energy conversion system. Fundamental study with a model system involving similar characteristics to living matter is one of the powerful approaches for understanding and application of such types of motion. In this study, we have studied the motion of aluminum float driven by the spontaneous flow at oil/water interface: The oil and water phases contain di(2-ethylhexyl) phosphate (DEHPA) and Fe2+, respectively, whereby spontaneous instability develops at the interface. Spontaneous flow is generated by a local compression of the adsorbed DEHPA layer by an interfacial flow, and the generated flow compresses the surrounding DEHPA monolayer to cause the subsequent flow. This suggests that spatiotemporal control of compression points rectifies the flow pattern, resulting in a regulated motion of a float.
As an example, a rectangular aluminum foil is placed in an arc-shaped triangular space as shown in Fig.1(a). The aluminum foil exhibits a reciprocal motion. This motion repeats itself many times. Such a motion was not obtained without the confined space. The motion of the aluminum foil results from the compression of DEHPA layer between the aluminum foil the wall, because the compression induces the next flow. As the other example, a ratchet gear made of aluminum foil is placed at the interface where powder are dispersed in the oil/water interface(Fig.1(b)). The ratchet rotated in one direction, that lasted for more than 10 minutes. This rotation may be considered to be caused by the compression of DEHPA layer around the aluminum foil assisted by the powder acting as a wall. These motion may lead to the creation of an innovative energy conversion system.

Graphite consists of many carbon sheets. A single layer of the carbon sheets is called graphene. Graphene is a material having good electrical conductivity and high mechanical strength. Industrial applications of graphene are expected in the wide range of fields. However, graphene is usually aggregated in solvents because of strong hydrophobic interaction. This causes the difficulty of the manipulation and the loss of electrical conductivity of graphene.
We previously succeeded in dispersing graphene using poly(3-hexylthiophene-2,5-diyl) (P3HT), which is the conductive organic polymer, in an organic solvent. However, P3HT dispersed graphene only in an organic solvent, which leads to large environmental concerns. Here, we aimed at the preparation of graphene aqueous dispersion using poly(3-(4-sulfophenyl)thiophene-2,5-diyl) (P3ST), which is a water-soluble polythiophene derivative.
P3ST is obtained by deprotecting neopentyl groups of poly(3-(4-neopentylsulfophen-1-yl) thiophen-2,5-diyl) (P3NST). We mixed graphite flakes with P3NST in chloroform and conducted ultrasonication for exfoliation and dispersion of graphene. After the removal of chloroform, the graphene-P3NST complex was heated at 220~230 °C for deprotecting neopentyl groups of P3NST. Then, we added water and redispersed the graphene-P3ST complex in water by ultrasonication. After the redispersion was left for 20 h, the supernatant was collected. The supernatant was diluted 2 times with water and its absorbance at 800 nm was measured to evaluate the dispersion of graphene.
Fig. 1 shows the absorbance at 800 nm of the dilute solution of the supernatant. The x axis shows heating time for deprotecting neopentyl groups of P3NST. The results showed that dispersion of graphene in water was improved by heating the graphene-P3NST complex. The deprotection of neopentyl groups produced sulfo groups in the polythiophene derivative and the sulfo groups would generate electrostatic repulsion between the graphene nanosheets in water to stabilize the dispersion.
Cu-free click reaction is one of the most famous reactions for conjugating DNA, proteins and cells. Recently, a Cu-free clickable surface is attracting as a platform of immobilizing biosubstances for preparing biosensors. In previous studies, inorganic substrates (e.g., silicone and glass) were used, and they needed surface activation by plasma and chemicals. In this study, we synthesized a Cu-free clickable functional polymer, and a Cu-free clickable surface was prepared on a plastic substrate by polymer coating without pre-treatment.
We synthesized a methyl methacrylate (MMA) and hydroxyethyl methacrylate (HEMA)-based backbone copolymer. Then, dibenzocyclooctyne (DBCO), a Cu-free clickable functional group, was conjugated to the hydroxy group of PMH to get a Cu-free clickable polymer, poly(MMA-r-HEMA-DBCO) (termed PMHD). PMHD was dissolved in ethyl acetate, and a plastic substrate was coated with the polymer solution and dried to prepare a Cu-free clickable surface. AFM and SEM observations revealed that dip-coated surfaces were flat compared with bare substrates.
We also synthesized an azide-conjugated fluorescent substance and estimated the amount of clickable moieties on the polymer-coated surface using Cu-free click reaction between the surface and the fluorophore. The surface density of Cu-free clickable moieties was proved to be 63 pmol/cm2 on PMHD-coated PMMA substrates. This meant that a Cu-free clickable surface was easily prepared on a plastic substrate by polymer coating. To investigate the potential of the functional surface as a platform of a biosensor, azide-conjugated oligo DNA was immobilized on the clickable surface. In consequence, 5.4 pmol/cm2 azide-conjugate oligo DNA was immobilized and hybridized with the complement DNA on the PMHD-coated functional surfaces.
In summary, we synthesized a functional polymer (PMHD), and succeeded in the preparation of a Cu-free clickable surface by simple polymer coating. The investigation of DNA immobilization revealed that the functional surface had high potential as a scaffold of biomatters.
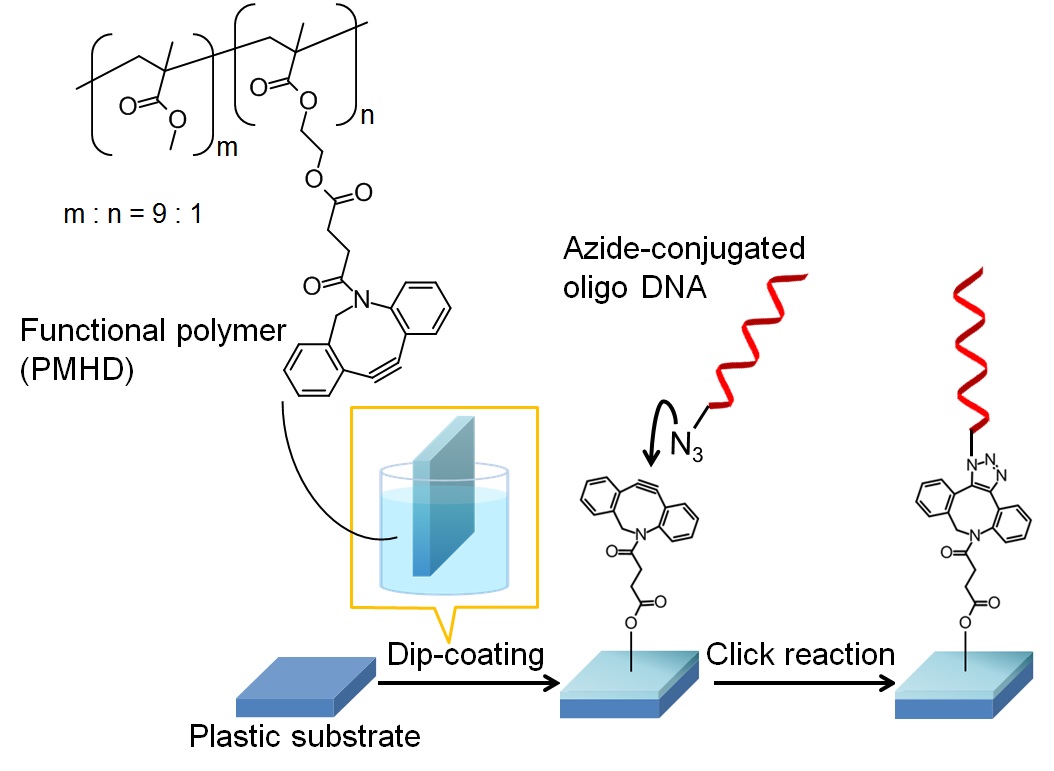
In recent years, separation membranes have attracted a lot of attention: that is widely applied to seawater desalination treatment, separation/recovery of exhaust gas, etc. The materials of the separation membrane are classified into inorganic or organic one, and the latter has generally advantages of low cost, low density and easy handling. In the widespread use, it is indispensable in our lives.
At present, a new preparation process using phase separation in liquid phase has been developed. The outline of this process is 1) selection of a polymer, its solvent and anti-solvent, 2) mixing of them, 3) emergence of phase-separation at specific compositions and 4) drying and formation of porous separation membranes. However, the formation mechanism of the membrane is not clear in the drying process and should be clarified for design of the membrane structure. In this study, poly(vinyl alcohol) (PVA), water and γ-buthyrolactone(GBL) were selected as ingredients for the membranes. Then, the concentration changes of mixed samples have been investigated in the formation of the porous membranes of PVA by the drying process.
As a first approach, preparation of the phase diagram for the three components system was carried out.When the concentration of the liquid sample changed by adding a component slightly, the turbidity was measured. The binodal curve was found by connecting of the turbidity points in PVA-GBL-water systems. The trace of concentration change in drying processes were drawn in the obtained phase diagram. The structures of PVA on the trace line was measured by SEM and DSC measurements were carried out. The relationship of drying rate and PVA structure was elucidated with the structural and thermal data.
Plastics have some practical characteristics; in recent years, the modification of plastic surfaces has received considerable attention for new application. The display of reactive functional groups on plastic surfaces is one approach of the modification of plastic surfaces and it is very important for adhesion and immobilization of biomolecules and polymers. In this study, we displayed amino groups and immobilized gold nanoparticles (AuNPs) on plastic surfaces. Preparation of AuNPs-modified surfaces will realize new applications such as fabrication of conductive plastic surfaces, immobilization of thiol compounds on a surface of a plastic substrate.
We synthesized a methacrylate-based copolymer containing amino groups in the side chains, in which the amino groups were protected with tert-butoxycarbonyl (Boc) groups. The protection groups of amino groups in this copolymer were hydrophobic, leading to the display of the amino groups on the outermost surface of a PMMA substrate after dip-coating. We deprotected the Boc groups and displayed amino groups on the surface of the substrate. Quantitation of the amino groups was carried out using a cleavable fluorescent compound. The amount of amino groups displayed on the surfaces was 10.3 pmol/cm2.
AuNPs, which were 13 nm in diameter, were immobilized on the plastic surfaces by simply immersing PMMA substrates in Au-colloidal solution for 30 minutes. The acrylic substrate displaying amino groups turned red after immersion (Fig. 1), comparing to the bare substrate and the substrate displaying Boc groups. We measured absorbance of these substrates after immersion at 520 nm, which was maximal absorption wavelength of Au-colloidal solution. Absorbance of the substrate displaying amino groups was the highest among the three different kinds of substrates. We conjectured that electrostatic interaction between amino groups and negatively-charged AuNPs led to the immobilization of AuNPs on the surface.
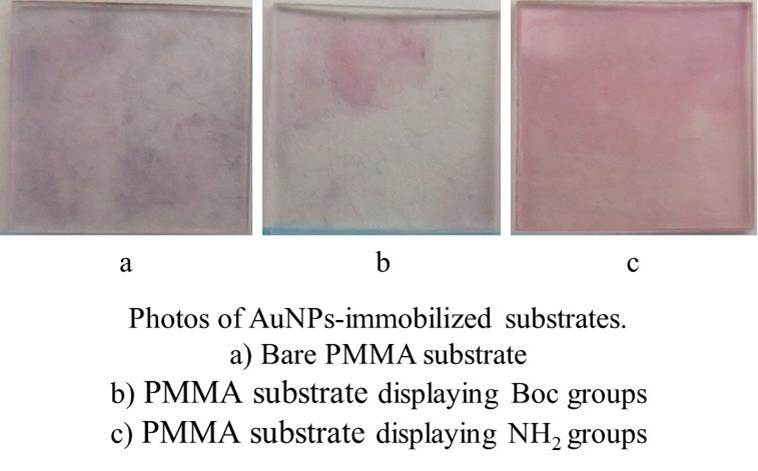
In drying of particle dispersion films, drying defects such as cracking or the like are caused by capillary forces acting between the particles. Previous studies have shown that cracks develop when the dry film thickness exceeds a critical thickness, referred to as critical cracking thickness (CCT)[1]. Polymeric additives can increase CCTs as they (i) adsorb on particle surfaces to induce steric repulsive forces between the neighboring particles, (ii) adsorb at air-liquid interface to reduce the surface tension, and thus the capillary forces, or (iii) show physical crosslinking between particles. However, few quantitative data are currently available to clarify how polymers suppress the propagation of cracks. In this study, we aim at investigating the effects of polymer adsorption on critical cracking thicknesses of drying particle dispersions.
We used Carboxymethyl cellulose (molecular weight: 1850000 g/mol, Dai-ichi Kogyo Seiyaku) as polymer. The sample dispersion containing TiO2 (diameter: 150 nm, concentration:15 wt%) and polymer was dried on a tilted hot stage. The dried film surface was observed by an optical microscope to find a position where cracks developed with a length of 30 μm or longer. The film thickness at that position was measured using a laser focus displacement meter to determine CCTs.
The results showed that CCT was nearly constant at polymer concentrations lower than the critical concentration of c* ~ 0.05 wt%, and agreed with that in polymer-free particle dispersions. At polymer concentrations above the critical value, on the other hand, the CCT monotonically increased with increasing the polymer concentrations. The adsorption measurements indicated that the onset of polymer adsorption on the particle surface emerged at the same polymer concentration, c*, indicating that crack propagation was suppressed by polymer adsorption on the particle surface.
References
[1] K.B.Singh and M.S.Tirumkudulu,Phys.Rev.Lett.98(21)(2007)218302
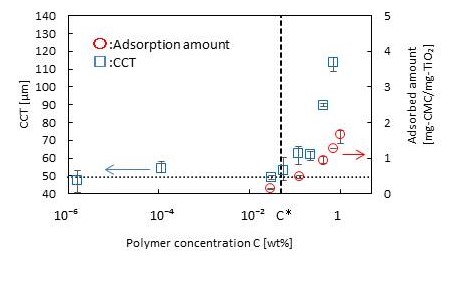
Shape of particles in dense suspension influences on the rheological properties. We focused on wrinkles generated by buckling as the periodic structure on the surface of elastomer particles, and evaluated the suitable method to control the obtained particle size and the shape of wrinkles.
Polydimethylsiloxane (PDMS) was used as the base material for wrinkled particles. The monomer and curing agent were mixed (10:1 (w/w)) to obtain the uncuring PDMS sol, and then the sol was mixed in a 15wt% polyvinylalcohol/H2O solution by the following methods, magnetic stirrer (ST), homodisper (HD), and homogenizer (HG). The PDMS droplets were filtrated with the porous membrane to fractionate the droplet size. In order to cure PDMS, the droplet suspension was heated at 80 °C for 3 hours. Finally, the surface of the PDMS particles were oxidized with a mixed acid solution to form wrinkle structure. The size of PDMS were measured from the image of optical microscope and/or dynamic light scattering.
The order of PDMS droplet size was roughly ST > HD > HG, and the particle size distribution was very broad from several 10 nm to above 100 μm. To separate out the particles with desired size, the HD pre-mixed PDMS suspension (1000 rpm, 5 min) was filtrated by using a shirasu porous glass membrane (pore size: 1 μm). The filtrated PDMS suspension shown a more narrow size distribution (d50 ~2.5 μm (Figure)). However, after surface oxidation of mixed acid, surface wrinkling on the filtrated PDMS particles was not observed by a SEM. Only PDMS particles of 15 μm or more in the diameter obtained from HD pre-mixed suspension were buckled into wrinkling. The wavelength of wrinkle was about 450 nm (d~15 μm), which tends to increase slightly with the particle diameter.
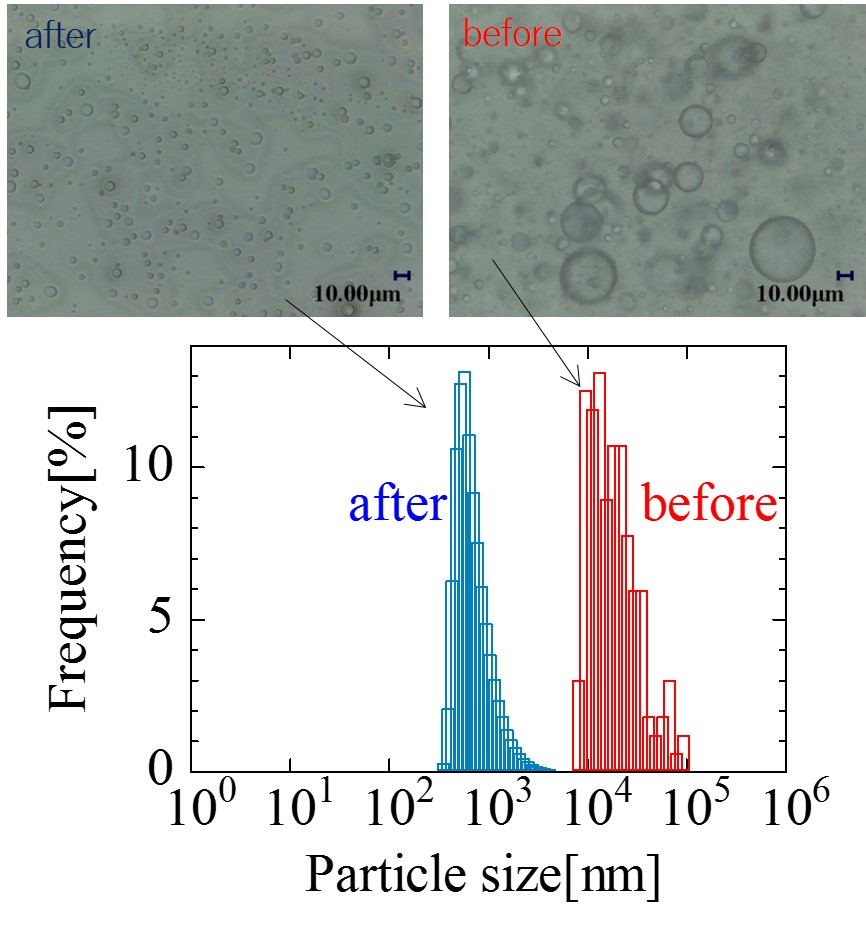
The organic luminescent material easily loses its luminescence once it is exposed to moisture, and the highly reactive cathode with low work function will be easily corroded by moisture and oxygen. To solve this problem, We demonstrate flexible moisture barrier film encapsulation for organic electronics using low temperatures process method. To realize the flexible barrier, the best practice is to make the barrier as multilayer structure. Low temperatures process applied the atomic layer deposition(ALD) method and sputtering method to deposit barrier films. Inorganic layers work as moisture barrier and very thin thickness provide the flexibility and planarization. The moisture permeation characteristics of the optimized Al2O3/SiNX multilayer films were finally measured using MOCON's WVTR measuring instrument and WVTR values lower than the detection limit of the measuring device (less than 5.0 x 10-5 g/m .day) were obtained.
This study is to investigate the optimal time of applying current assisting during employing hot-pressing Bi-Sb-Te films to improve the thermoelectric properties. First, an average 5 um of grinded P-type of Bi-Sb-Te powers was used to formulate a slurry paste to coat on a polyimide substrate for the current assisted hot pressing. It shows that the resistivity decreases with increasing the current density, and then increases with further raising the current density. The main reason is speculated that increasing the electric driving force at low current density can eliminate defects by reducing the obstacles of carrier transmission and formatting Sb-rich phase to form a bridge between grains. However, the excessive current density will generate stronger electric driving force causing a large amount of joule heat to increase the resistivity. Also, the case of “hot pressing and current assisting simultaneously” or the case of "current assisted hot pressing after completion of hot pressing" cannot achieve the best improvement of the thermoelectric properties. Analyzed by surface reaction method, the best thermoelectric efficiency can be obtained at 1000 A/cm2 with 3 min hot pressing followed by 7 min current-assisted hot pressing. Applying hot pressing ahead can increase contact area between particles to make current density distribution more uniform, which can generate less extra joule heat and larger electric driving force, leading to further reducing resistivity to 1.04 mホゥ·m and increasing power factor to 2880 μW/K2m. Compared with the case of hot pressing and current assisting simultaneously for 10 min and the case of current-assisted hot pressing for 7 min after hot-pressing for 10 min, the power factor increases 34% and 20%, respectively.
An innovative bio-functional lenses can control multiple surface properties independently for bio-medical applications. The bio-functional lenses was fabricated based on a chemical vapor deposition encapsulation process using functionalized poly-p-xylylenes. Our device exhibits excellent biocompatibility, high transparency, heat resistance and stable in vacuum. The bio-functional lenses applied in many area, such as artificial crystal or cell incubator. Here we reported a method for the fabrication of bio-functional lenses which constructed via vapor deposition of poly-p-xylylenes on template and shape is controlled by oil. Because the lenses constructed by liquid, it is softer than solid that apply in artificial crystal. On the other hand, the lenses have some characteristic such as stable in vacuum, biocompatibility, and perfect curvature. Another research, we try to put Human Adipose Stromal Cells(ASC) in the lenses which is encapsulated by oil and observed in Transmission electron microscope (TEM). The advantage of lenses is high transparency that easily observed in TEM and durable in the environment of vacuum. We build an excellent cell incubator so that we can observe life phenomenon in TEM instead of killing cells.
Perovskite solar cells have been intensively studied because they have high efficiency over 20%1 and are fabricated by a low-cost wet process. The ideal cell structure is a simple planar type with a multi-layer of substrate/electrode/electron transport layer/perovskite power generation layer/hole transport layer/electrode. Titanium dioxide (TiO2) is used as a compact blocking layer as well as an electron transport layer. The underlying TiO2 layer would also play a role in determining the structure of a perovskite layer. To improve the efficiency of the solar cell, it is important to control the structure and physical properties of the TiO2 layer. The ultrasonic spray method is one of the wet coating processes. It enables to control film structure by nuclear growth on the substrate and to deposit large-area and continuous film, having advantages of applicability of the Roll to Roll process upon scale-up to mass-production. The aim of this study is to analyze the relationship between coating process, film structure and physical properties of TiO2.
Titanium bis acetylacetonate diisopropoxide (Ti(acac)2(OiPr)2) and titanium tetrachloride (TiCl4) were used as soluble precursors in ethanol. The ultrasonic spray method was used to deposit TiO2 films on two different types of SnO2 films. The structure of the TiO2 films was investigated by X-ray diffraction and X-ray absorption spectroscopy. In the case of Ti(acac)2(OiPr)2 solutions, TiO2 films had anatase and rutile structures with (101) orientation, while the films deposited with TiCl4 solutions showed only a rutile structure with (101) orientation. The local epitaxial growth of rutile TiO2 on rutile SnO2 structures would be the origin of the observed rutile (101) orientation. We will discuss the effect of the structure of the TiO2 films on the physical properties of perovskite solar cells.
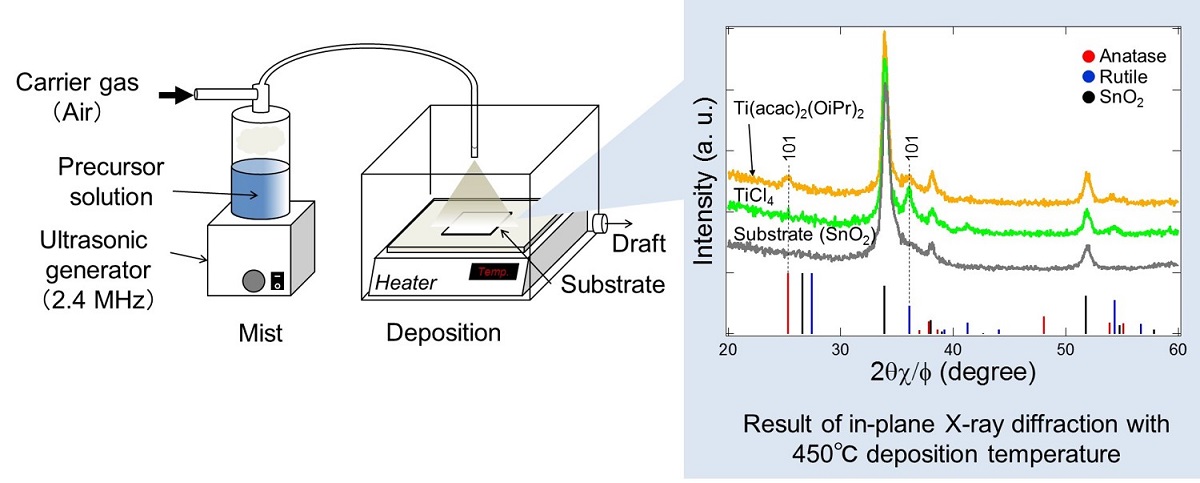
Fluorinated compounds often exhibit a very low surface energy, and these can be used as antifouling and antifogging agents. However, high F-content compounds are generally expensive and bioaccumulative. Our previous research found that non-fluorinated surfactants having trimethylsilyl groups decreased aqueous surface tensions as low as a short or middle fluorocarbon-tail surfactant can (i.e. ~22 mN/m). To develop fluorine-free water-repellent materials comparable to fluorinated one, this study synthesized 3,4-propylenedioxythiophene monomer having 3-trimethylsilylpropyl group (ProDOTSiMe3, Fig. 1) and obtained poly(ProDOTSiMe3) films on gold surfaces by electrodeposition of the monomer.
Electrodeposition was carried out by placing a gold plate (working electrode), a glassy carbon electrode (counter electrode), and SCE in a glass cell containing 10 mL dehydrated acetonitrile solution with 0.1 M tetrabutylammonium perchlorate and 0.01 M ProDOTSiMe3. Water repellency and morphology of the surfaces were also studied. SEM observation clarified that the gold surfaces after electrodeposition were covered with dense poly(ProDOTSiMe3) nanofibers with a large roughness. Water contact angle on the gold surface was 141.6 °ツア 4.7 °, similar to the values 136.8 °and ≥ 150 °for 3,4-propylenedioxythiophene monomers having two isopropyl-terminated alkyl chains[1]. Interestingly, the poly(ProDOTSiMe3) surfaces exhibited parahydrophobicity due to the wenzel-state.
References
[1] Thierry Darmanin, Claudio Mortier, Julian Eastoe, Masanobu Sagisaka, Frederic Guittard., RSC Adv., 2014,4, 35708–35716
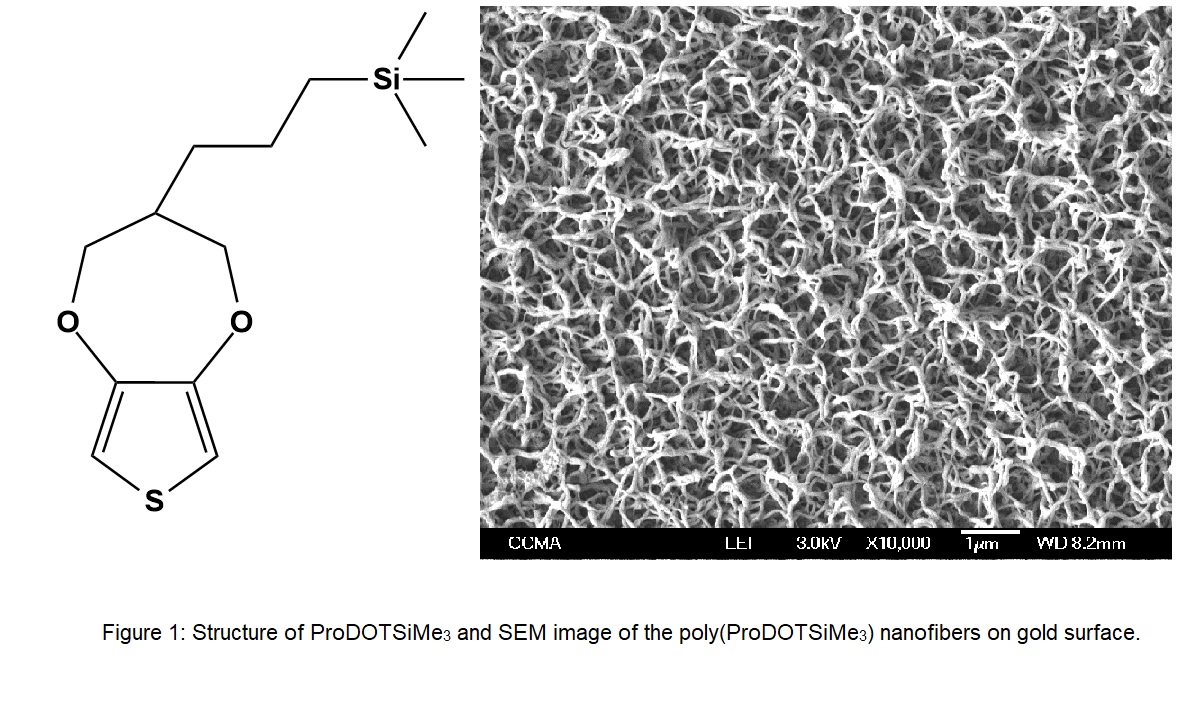
A probe needle for a probe card which is employed in a current energization inspection of an integrated circuit chip in a semiconductor wafer manufacturing process has been made of tungsten, rhenium tungsten, beryllium copper, etc. Particularly, tungsten is a material which is excellent in wear resistance and the excellent toughness is popularly used. However, the contact resistance is raised and the liability of probe card deteriorates on the inspection because tungsten is easily oxidized due to the frictional heat which is generated by the frictional wear or the generation of heat. In order to solve this problem, it is desirable to use gold or platinum material, which is strong in corrosion resistance, as a probe needle. However, in case of gold, it cannot be used in contact with the pad due to its ductility. In addition, there is no economical efficiency because the process cost is increased.
In this study, we platted on the surface of tungsten needle with 1 ~ 5 um of gold, platinum, and nickel to prevent the oxidation of tungsten needle by frictional heat which can be used sufficiently as a probe from being corroded by heat. The undercoat was conducted with nickel, and then, gold, platinum, or nickel was plated. The change of electric resistance after plating was examined. Because the needle thickness is very thin, the over-plating occurred at the tip of tungsten needle. To reduce over-plating, new zig for needle was invented and plating conditions were optimized.
Figure1. The microscope cross-sectional images of bare tungsten needle, the plated tungsten needles with nickel, gold, platinum.

High-chromium cast iron, which is a wear resistant material, is a ternary white cast iron generally containing 7% or more of Cr. It forms many carbides of high hardness, for example M23C6 and M3C, M7C3 (M = Fe, Cr)(1).Therefore, it is used as a material for rolling rolls used in rolling processing and slurry pumps for slurry liquid transfer. However, the rolling conditions are becoming more severe in terms of energy saving and quality improvement in rolling processing, and the slurry pump has problems of wear and corrosion due to high concentration slurry liquid. In order to cope with the current situation, it is important to change the solidified structure by adjusting the Cr and C content, in order to improve the low toughness, corrosion resistance, and wear resistance. Changes in the composition and cooling rate that affect the solidification structure are factors related to the mechanical properties of cast iron. Direct observation of the solidification process in which carbides and base structures appear is thought to be important because it leads to the elucidation of the growth mechanism and can be expected to control the size, amount and formation position of the crystal. Direct observation of metals under high temperature is carried out to clarify the growth mechanism of the metal structure. For this purpose, it is necessary to use a laser microscope which is not affected by the radiation emitted from the molten metal. In this research, our purpose to establish the observation condition of the growth process of base structure and carbide using laser microscope and to elucidation the formation of structure
(1)Tomoo Sato, Taiji Nishizawa, Joo Ishihara, hardness of carbides in iron and steel, Journal of the Japan Institute of Metals, Vol. 23, No. 7, pp. 404-405, 1958