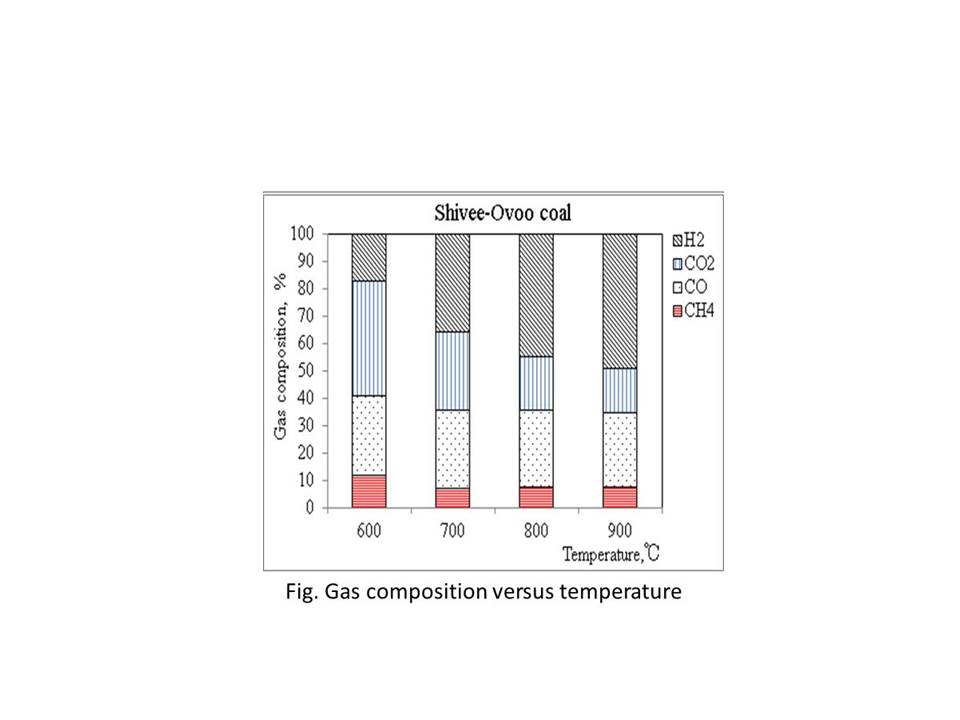
In this study, pyrolysis of Mongolian three different brown coals was investigated in the fixed bed reactor and fluidized bed reactor. In the fluidized bed reactor, the porous alumina and quartz sand were used as bed material for studying the influence of bed material. Size range of bed materials (quartz sand and alumina) was -250μm +212μm and the gasifier temperature between 600 and 900oC. The main components of the obtained syngas were CO, H2, CH4 and CO2, which were measured by Gas chromatograph. The gas and tar yields were reported in more detail at temperature from 600oC to 900oC. Also, the composition of char was determined at given temperatures.
The coal and gas fired power generation are defined as significant power sources, however it is necessary to reduce CO2 emissions from thermal power generation. In this session, I will introduce the development trends and prospects of next-generation thermal power generation technology in Japan.
Spontaneous ignition of coal stockpile is a serious economic and safety problem.
Theorefore, coal oxidation has been received great attention and many studies has been carried out.
Although coal oxidation at actual stock yard proceeds under the condition of low temperature and humid atmosuphere, almost all of previous study has been carried out under the condition of high temperature (>100 °C) or dry atomosphere.
In this study, coal oxidation under the condition of low temperature and humid atmosphere was investigated.
CO and CO2 were the major products and the formation rate of CO2 was larger than that of CO.
The heat generation from coal was also observed and that was followed by the formation of carbon oxides.
Therefore, it was considered that the main factor of heat generation was oxygen absorption which is initial step of coal oxidation.
Heat generation rate under dry atmosphere was almost the same as that under humid atmosphere.
So water would have little influence on heat generation at this condition.
CO formation rate under the dry atmosphere also almost the same as that under humid atmosphere.
On the other hand, CO2 formation rate under the dry atmosphere is much smaller than that under the humid atmosphere.
As a result, water has pronounced effect on CO2 formation reaction.
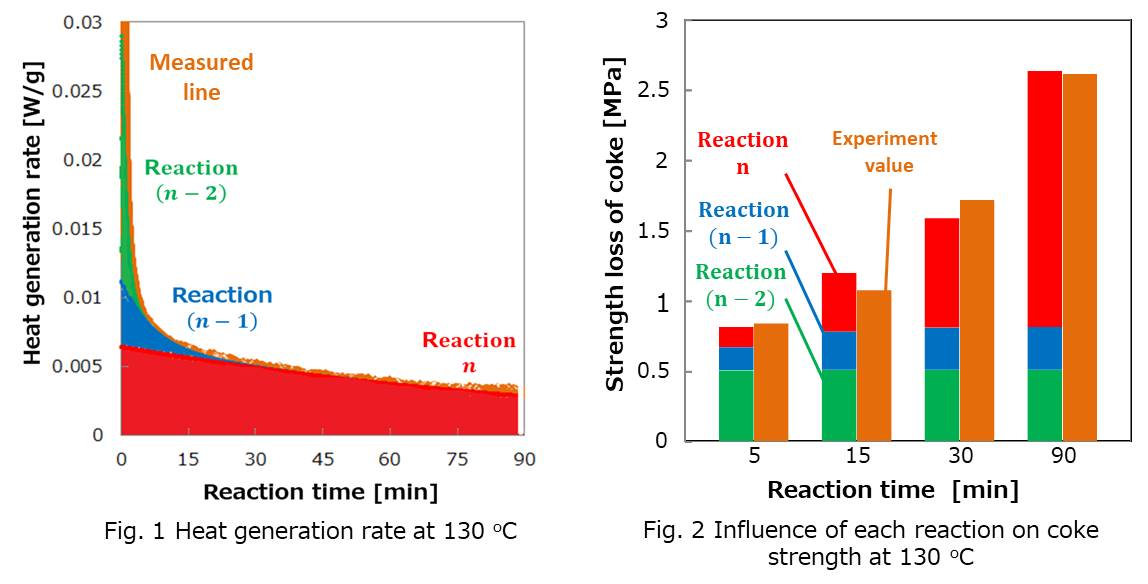
Coke serves as fuel, reductant and support material to allow continuous flows of gas and liquid in blast furnace of ironmaking industries. Over the last few decades, since the scale and production ability of blast furnace kept increasing, mechanical strength of coke became more and more important. However, when raw coking coal is exposed to air, it would be weathered, which leads the coke made from it can not obtain enough strength to meet the requirement of ironmaking industries. It is necessary to investigate the influence of weathering on the coke strength quantitatively.
Oxidation at relatively low temperature is believed to be one of the main reasons for weathering. In this study the influences of the oxidation level of raw coal at low temperature on the softening and melting ability of raw coal and the mechanical strength of coke were investigated from the viewpoint of oxidation reaction rate.
Since the weathering takes several months, simulated weathered coal was prepared by heating coking coal in air at 120 oC to 150 oC, where oxidation reactions were accelerated, for 5 to 90 minutes. Then coke was prepared from the simulated weathered coal. The coke obtained had less mechanical strength as the treatment temperature and time increased. The heat generation rate in the oxidation reaction at each temperature was measured by differential scanning calorimetry. Reaction rate analysis showed that the oxidation reaction of raw coal could be divided into three parallel first-order reactions (Fig.1). It was found that in the long run, the reaction which has the lowest reaction rate constant had the most significant influence on coke strength (Fig.2).