In this work, cerium-zirconium oxides particles were synthesized by solvothermal and hydrothermal method at low temperature in order to be utilized as catalyst in the delignification of wood biomass. The experiments were conducted at temperatures of 150oC – 250oC and pressures of 2 MPa – 5 MPa for 2 h in an autoclave made of SUS 316 with internal volume of 100 mL. Water, mixed of water and ethanol (70/30 vol/vol), and mixed of water and ethylene glycol (70/30 vol/vol) were used as solvent, while Ce(NO3)3 and ZrO(NO3)2 with 0.06 M concentration were used as precursor. The synthesized products were dried at 60 oC for 6 and then calcined at 500 oC for 6 h. The particle products were characterized using SEM and XRD. Furthermore, the particle products were used for hydrothermally delignification process of wood biomass. The addition of cerium-zirconium particles dramatically increased % lignin removal of rapeseed wood up to 97.6%. Based on the result, cerium-zirconium oxides particles are effective for pre-treatment of wood biomass in bio-refinery application. Moreover, cerium-zirconium oxides may reduce the use of chemicals in the delignification process.
Plant derived biomass has been focused as chemical feedstocks as well as an alternative fuel. Cellulose can be converted to glucose and cellooligosaccharides, which are ingredients for chemicals, pharmaceutical, dietary foods etc. However, due to its high crystallinity caused by inter- and intra-molecular hydrogen bonding it is not easy to convert to monosaccharide and oligosaccharides efficiently. Thus, a new efficient process to convert cellulose to monosaccharide and oligosaccharides is demanded. Hydrothermal conversion is one of the promising methods. In this study, filter paper as a cellulosic sample was hydrolyzed using a batch reactor under hydrothermal conditions. The effects of temperature, alkali species and the concentration on product distribution, reaction rates and the degree of polymerization (DP), crystallinity index (CrI) of hydrolysis residue were investigated.
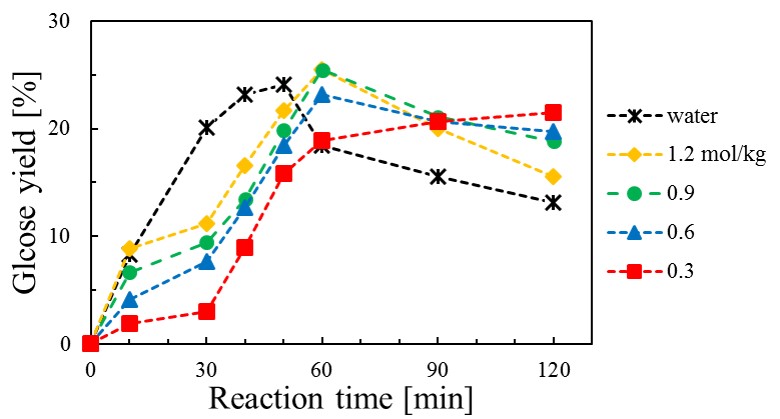
In a batch reactor (volume 3.5 mL) ca. 0.05 g- filter paper and ca. 2.5 g- alkali solution were loaded at room temperature. Alkalis used were methylamine, dimethylamine, trimethylamine, ethylamine, diethylamine, triethylamine, propylamine, n-butylamine, s-butylamine, t-butylamine, pyridine, sodium hydroxide, aniline, benzylamine and ammonia. The hydrolysis experiments were carried out from 513 to 553 K. The product yields were measured by high performance anion exchange chromatography. The major products were glucose, cellobiose, fructose, levoglucosan and 5-hydroxymethylfurfural. Total organic carbon in the solution was also measured by a TC analyzer. The hydrolysis residues were analyzed by X-ray diffraction and CrI. The CrI values were calculated from the equation of segal1).
Fig.1 shows the effect of pyridine concentration on the glucose yield at 533 K. Glucose yield decreased after 60 min at pyridine concentrations above 0.6 mol/kg. We conclude that further decomposition products such as levoglucosan and 5-hydroxymethylfurfural were produced after 60 min.
1) L. Calvo et al., Ind. Eng. Chem. Res., 41, 6503 (2002).
Keratin is a protein contained in human hair. The keratin contains 17% of the sulfur-containing amino acid cysteine, which could form disulfide bridges. Recently, low-molecular-weight keratin peptides are receiving a lot of attention in cosmetics and hair repair agents because of their high affinity to the skin and hair. At present, the low-molecular-weight keratin peptides are produced by the enzymatic hydrolysis of the keratin raw materials such as feathers. However, the enzymatic hydrolysis of the human hair keratin is difficult because the cuticle keratin protects the surface of the human hair.
In this work, we conducted the hydrothermal treatment of the human hair to hydrolyze keratin proteins. Since the hydrothermal treatment uses only water, the separation of the enzyme and the peptide is not needed and the peptide is safe as a hair repair material. The influence of reaction conditions (temperature and treatment time) on the molecular weight was studied.
0.1g of human hair and 1 mL of water was loaded into a stainless 316 tube reactor (inner volume 6 cm3). The rector was immersed into a sand bath at a temperature of 150, 180, and 250 °C. After 5~30 min, the reactor was removed from the sand bath and rapidly quenched in a water bath to room temperature. After solid and liquid products were separated by a filtration, the molecular weight of the products in the liquid phase was measured by the size-exclusion chromatography (SEC).
The low-molecular-weight keratin peptides could be obtained from human hair by the hydrothermal treatment. The low-molecular-weight keratin peptide with a peak at around 3.0 kDa was obtained at 150 and 180 °C. At 250 °C, the peptide with a peak at around 1.0 kDa was produced. The effect of the molecular weight of the keratin peptides on the repair capacity is also studied.
Chitin is the second most abundance resources after cellulose and mainly contained in the exoskeletons of crustaceans such as crabs and squid pens. Chitin nanofiber (NF) has attracted research interest in medical materials and healthy foods in recent years because chitin NF has large specific surface area and high biocompatibility. Furthermore, there are two types of chitin depending on crystalline structure. α-Chitin has antiparallel molecular chains with hydrogen bonds between molecular chains. β-Chitin has parallel molecular chains without hydrogen bonds between intermolecular sheets. Therefore, nanofibrillation of β-chitin is easier than that of α-chitin because of the absence of hydrogen bonds. In our previous report, we have prepared β-chitin NF dispersions with high viscosity and high transmittance from β-chitin powder; however, α-chitin could not be disintegrated into such NF dispersion due to many hydrogen bonds. In addition, we have obtained self-sustaining hydrogels from β-chitin NF dispersions by hydrothermal treatment; however, it is difficult in the case of α-chitin. If the gelation of α-chitin NF can be achieved, mass production for commercialization will be realized because α-chitin is more abundant in nature than β-chitin. Thus, it is important to establish the gelation method of α-chitin NF by hydrothermal treatment.
In this work, we obtained the α-chitin NF dispersions with high viscosity and high transmittance by reducing degree of N-acetylation and adding acids. The disintegration of α-chitin powder into NF was conducted with a water jet machine (Star Burst System, Sugino Machine Co, Ltd, Japan). The prepared α-chitin NF dispersions showed high viscosity and high transmittance. In addition, the hydrothermal treatment of the prepared α-chitin NF dispersions was conducted at 160 °C for 60 min by using a sealed reactor. After the hydrothermal treatment,α-chitin hydrogels with high mechanical strength could be obtained.
Supercritical carbon dioxide (scCO2) dyeing for textile and polymer processing is considered as a green and energy-saving process for view-points of environmental friendly and sustainable development. We developed a compact apparatus dyeing for plastic buttons of acrylic, polyester, nylon, and casein in SC-CO2 and carried out dyeing plastic buttons with C. I. Disperse Red 22. We could successfully dye the plastic buttons without cracks, bubbles, and black spots over the temperature ranges of 313.15 to 328.15 K and pressures of 8 to 14 MPa for acryl, the temperatures of 383.15 to 398.15 K and pressures 10 to 16 MPa for polyester, and 353.15 to 383.15 K and pressures of 8 to 14 MPa for casein with dyeing treatment time of 30 to 60 min, and the temperatures of 383.15 to 403.15 K and pressures of 21 to 29 MPa for nylon with dyeing treatment time of 60 to 100 min. The dyeing color depth of dye sorption in the buttons were measured by a spectral color difference meter. Also we measured the solubility of C. I. Disperse Red 22 over the temperature and pressure ranges of 353.15 to 398.15 K and 15 to 25 MPa. The K/S values for plastic buttons were correlated by a regression model, which was formulated by combining Fick diffusion equation with Henry-Langmuir type of surface adsorption of buttons in the present work. Using the model, we examined to represent the experimental K/S in terms of temperature, pressure, density, solubility of dye, and dyeing time. Finally, we compared K/S value by the regression model with those obtained by a multiple regression model expressed in terms of temperature, pressure, density, solubility of dye, and dyeing time and the range analysis of experimental design method previously reported.
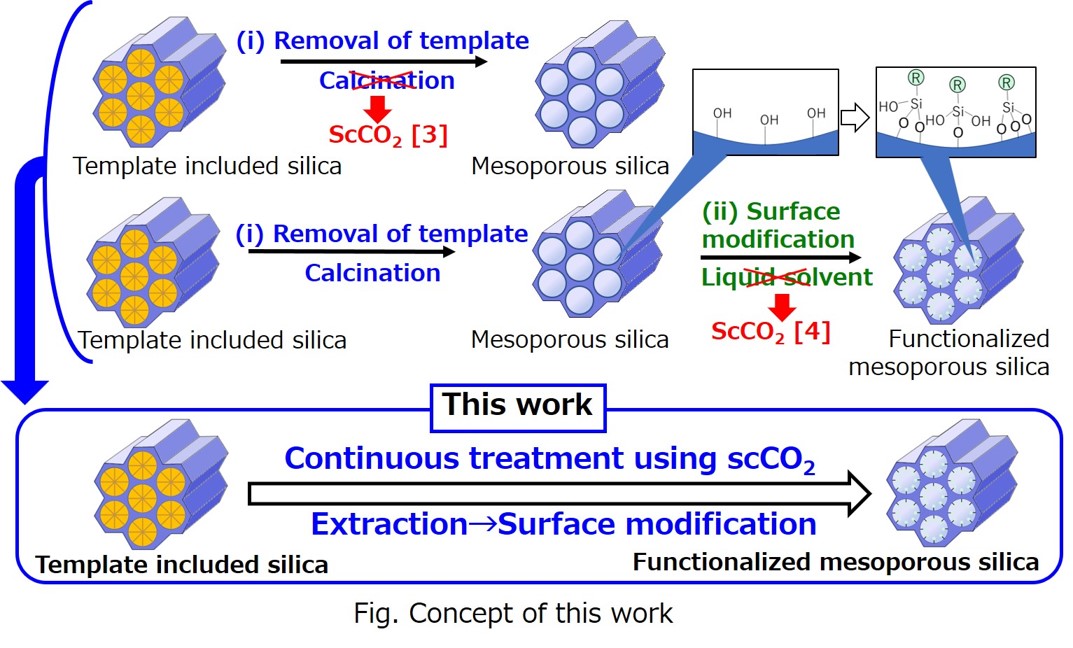
Mesoporous silica materials such as MCM-41 [1] and SBA-15 [2] have been widely used in various applications because of its uniform pore structure, large pore volume, and large specific surface area. Mesoporous silica materials are generally prepared by removing template agents by calcination processes from the template included silica (step (i)). Then, functionalized mesoporous silica can be obtained by the surface modification of silanol groups on the surface of the material using silane coupling reagents (step (ii)) with liquid solvents. However, these conventional methods have some drawbacks such as the possible shrinking of pore structures, disappearance of silanol groups, and low penetrability of solvents into the mesopores.
To overcome these drawbacks, in the present study, the novel preparation method of functionalized MCM-41 mesoporous silica was developed using supercritical carbon dioxide based on our previous works [3, 4], which can achieve continuous treatment of extraction of the template (step (i)) and surface modification of mesoporous silica (step (ii)). The functionalized MCM-41 prepared by the continuous treatment method demonstrated more uniform pore size distribution and higher amount of modification by silane coupling reagents in comparison with the conventional methods.
References
[1] C.T. Kresge et al., Nature, 359 (1992) 710-712.
[2] D. Zhao et al., Science, 279 (1998) 548-552.
[3] I. Ushiki et al., Kagaku Kogaku Ronbunshu, 37 (2011) 512-517.
[4] I. Ushiki et al., Kagaku Kogaku Ronbunshu, 38 (2012) 391-396.
Spent coffee ground (SCG) is regarded as a food waste. However, SCG contains various kinds of high-value phytochemicals such as diterpenes, polyphenols and lipids. Diterpenes present beneficial effects towards human health. Diterpenes can protect skin from the harm of UV, and they are anticarcinogenic, anti-inflammatory and antioxidant. These features of diterpenes make them valuable in both cosmetics and pharmaceutical industries. In this research, we extracted cafestol and kahweol from SCG using supercritical CO2 with ethanol. To maintain stability of the extracts and also to increase their solubility and dissolution rate in water, we studied the micronization of extracts containing cafestol and kahweol with PVP using supercritical CO2. As cafestol and kahweol are sparingly soluble in water, combining it with PVP (water-soluble polymer) and transforming it into small particles can improve the solubilities in water to enhance their absorption in human body. The main objectives of this work are to extract cafestol and kahweol from SCG and to micronize it with PVP using supercritical CO2. The response surface methodology (RSM) with Box-Behnken design was employed to study the effects of extraction parameters on the extracts. The ranges of three parameters are temperatures (40-60 °C), pressures (10-30 MPa), and ethanol flow rates (0-0.5 ml/min). The predicted optimum total diterpenes amount is 65.81 mg/g-extract at temperature of 80 °C, under operating pressure of 30 MPa, and with the ethanol flow rate at 0.3 ml/min. The micronization process was carried out by supercritical anti-solvent process under 40 °C, 25 MPa, CO2 flow rate (15 ml/min) and solution flow rate (0.25 ml/min). Cafestol and kahweol were successfully encapsulated into PVP particles with diameters ranging from 0.25 - 0.05 μm.
Bio-oil is often extracted from biomass by liquid solvent or supercritical carbon dioxide. However these extraction methods have several disadvantages. Liquid solvent extraction using hexane can be operated at an atmospheric pressure, but the extracted oil includes the impurities such as phospholipid and wax owing to the strong solvent power of hexane. On the other hand, supercritical carbon dioxide can extract bio-oil without impurities, but it requires high pressure condition. In this study, the expanded hexane with carbon dioxide is examined as a new extraction solvent. This solvent is diluted liquid hexane with carbon dioxide and it has less solvent power and higher selectivity of neutral lipid than normal liquid hexane does. Therefore the expanded hexane is expected to dissolve nonpolar oil and not to dissolve polar oil such as phospholipids.
The objective of this research is the development of new extraction technique of bio-oil from biomass using expanded hexane with carbon dioxide, where rice bran is used as a target biomass. The extraction was carried out at 25 oC, 4-5 MPa and 0.8-0.95 of molar fraction of carbon dioxide in expanded hexane with carbon dioxide. At 25 oC, 5 MPa, 0.86 of molar fraction of carbon dioxide, the oil yield was 1.2 times greater than that with normal liquid hexane because of the low viscosity and the high permeability of expanded hexane. Furthermore the phosphorus concentration in extracted oil was 6 ppm. It was very low level, comparing with that in rice bran, approximately 230ppm. This was because the solvent power of expanded hexane to neutral lipid decreased a little and that to polar components decreased much. As the results, expanded hexane with carbon dioxide could extract the phosphorus-free bio-oil with high yield under mild conditions.
Strawberry is one of the most popular fruits in Japan and its production is about 160 thousand tons per year. The leaf of strawberry is dumped after harvest. However, the leaf has high antioxidant property. Therefore, the extraction of antioxidants from the leaf of strawberry is expected as a production of new healthy food and effective utilization of biomass. In this study, the leaf of strawberry was extracted with a mixture of supercritical carbon dioxide and water that are non-toxic and harmless solvent.
The extraction experiments were conducted with semi-batch extraction system. At first, the leaf was introduced in the extractor, and then carbon dioxide and entrainer was supplied to the extractor. The extraction pressure was controlled with a back pressure regulator. The extract recovered was dried and weighted. The antioxidant capacity was evaluated by DPPH (2,2-diphenyl-1-picrylhydrazyl) radical-scavenging method on the basis of the amount of 2,6-di-tert-butyl-p-cresol (BHT), and the total phenolic content was evaluated with Folin-Ciocalteu method on the basis of the amount of gallic acid (GAE). The effect of entrainer was evaluated at 308 K and 20 MPa. The existence of water as an entrainer gave higher antioxidant capacity and the amount of total phenolic content of the extract than those in the absence of entrainer. At 20 MPa, antioxidant capacity and amount of total phenolic content tended to increase with increasing temperature. The extraction of antioxidants enhanced in high temperature region. At 313 K, the antioxidant capacity and the amount of total phenolic content tended to decrease with increasing pressure. The optimal condition for antioxidant capacity and the amount of total phenolic content was at 313 K and 20 MPa up to 333 K and 30 MPa.
Recently, attention is now focused on effective utilization of biomass, which is abundant in resources as a substitute resource for fossil resources. It is desirable to use woody biomass which is non-food biomass since the population is increasing, the use of food-based biomass may have new food problems. However, it is difficult to convert woody biomass to useful substances, and because high temperature processes are frequently used, a problem as high energy cost occurs. In recent years, therefore, attention is focused on chemical conversion of woody biomass using supercritical fluid technology. Based on the above background, attention was paid to tea fruit residue which is waste that don't cause food competition and capable of extracting useful components such as lipids or fatty acids in the previous research. The purpose of this work was to search conditions for high yield of lipids by extracting useful components from tea fruit residue on supercritical carbon dioxide. In supercritical carbon dioxide extraction, the effects of pressure, temperature and flow rate of carbon dioxide on the extraction yield of oil and lipid class were investigated.
Fluorochemicals have various unique characteristics [1] such as heat, chemical and non-adhesiveness/water repellency. Thus, fluorochemicals are used for various products to apply these characteristics. However, most of fluorochemical wastes are disposed by landfill and incineration above 1000 °C because of C-F bonds having high bond energy and electrical stability [2]. Prior to the disposal fluorochemical wastes should be made because toxic compounds could possibly be emitted in the course of their disposal. Namely, the incineration emits toxic compounds produced such as gaseous hydrogen fluoride, and the landfill elutes fluorine in environment [3]. Thus, a new effective defluorination process has been demanded. Many efforts have been made to remove fluorine from fluorochemical materials: by defluorination under hydrothermal conditions [3], electrochemical [4], and photocatalytic methods [3]. Our previous research found that the addition of ammonia and ammonia or sodium hydroxide were effective for hydrothermal dechlorination of PVC [5]. The objective of this study was to demonstrate the effectiveness of using aqueous basic solution in the hydrothermal defluorination of 4-fluoronitrobenzene. Certain amount of 4-fluoronitrobenzene and a basic aqueous solution were loaded in a small bomb-type bath reactor, and the reactor was heated in a molten salt or oil bath. Various basic species were tested, and sodium hydroxide was one of the most effective among them. The conversion rapidly increased in the early stage, and thereafter did slowly. The reaction rate will be discussed in the presentation.
[1] Maruzen Publishing Co., Ltd., Handbook of Chemistry: Applied Chemistry, 6th ed., 757 (2003)
[2] Moriwaki H. et al., Env. Sci. Tec., 39, 3388 (2005).
[3] M. Nozoe, et al., Env. Eng. Res., 43, 105 (2006)
[4] C. Schaefer, et al., Env. Sci. Technol., 52, 10689 (2018)
[5] K. Hashimoto et al., J. Mat. Sci. 43, 2457 (2008).
This study reports the separation of raw petroleum pitch materials by applying the supercritical fluid technology. The aim is to obtain the mesophase pitch, which is an important intermediate compound for various applications. Those applications include the battery manufacturing and artificial carbon materials. It is required to remove the low molecular weight and complex polycyclic aromatic hydrocarbons (PAHs) compounds from the raw pitch before the production of mesophase pitch. The purposes of this research are: (1) to develop a cleaner separation method for PAHs by using the supercritical fluid technology, and (2) to develop a relatively simple analysis scheme on determining the molecular weight and polydispersity by using the gel-permeation chromatography (GPC) with dual RI and UV detectors.
The separation process included two parts. The first part was the supercritical carbon dioxide extraction of the raw pitch sample. A stage-wise supercritical extraction process was investigated. Three extraction stages were operated at different temperatures. The second separation part used the residual pitch after the above extraction as the input materials. The subcritical anti-solvent (SAS) treatment was employed. In this SAS process, the relatively lower molecular weight part of the residual pitch was further removed. The final carbon dioxide insoluble pitch was obtained by using modified SAS equipment and novel experimental procedures. This final product was examined for its molecular weight and polydispersity. This final residual solid was ready for the next thermal condensation polymerization process. The most appropriate SAS operation condition was reported in this presentation.
A simple and effective analysis approach was reported in this study. The GPC with dual RI and UV detectors were investigated. A satisfactory calibration curve was obtained to determine the molecular weight after each separation stage in this study. The consistency between this analysis result with that from the complex NMR data was demonstrated.
Utilization of microfluidic systems for supercritical CO2 (ScCO2) extraction from the aqueous solution has gotten much attention to be applied for wide industrial fields. This is because the extractions in the microfluidic systems can achieve the high heat and mass transfer rates between ScCO2 and water phases, consequently improving the extraction efficiency. In the microfluidic system, understanding of the fluid flow patterns can be an important approach to clearly explain the extraction behaviour and to design the ScCO2 microfluidic system.
In this study, flow patterns of ScCO2-water and ScCO2-ethanol binary systems, and ScCO2-ethanol-water ternary system, are experimentally visualized in 1 mm of hydraulic diameter of window-square-channel. The flow patterns of the fluids after a micro-swirl-mixing technique are captured under different temperatures (32.0 to 60.0) °C and pressures (8.0 to 20.0) MPa. At near critical condition of ScCO2 (32.0 °C and 8.0 MPa), the mixture of ScCO2 and water formed a stable slug-flow. The flow pattern for ScCO2-ethanol system forms a slug-annular flow. The ternary system of ScCO2, ethanol and water resulted in the unstable slug-flow near the critical point (See Figure). In addition, at all other studied conditions, stable slug-flow with water slug formation are observed under ScCO2-water binary system. However, by changing the temperatures and pressures, different flow patterns are generated under ScCO2-ethanol binary and ScCO2-ethanol-water ternary systems. Wavy-flow, unstable slug-flow and single-phase flow are observed under ScCO2-ethanol binary system. While, ScCO2-ethanol-water ternary system forms the slug-annular-flow, wedging-slug-flow, unstable slug-flow and stable slug-flow at various temperatures and pressures. The formation of these flow patterns can be explained with respect to the phase equilibrium diagrams of ScCO2-ethanol binary and ScCO2-ethanol-water ternary systems.

Supercritical CO2 (scCO2) extraction is suitable for the separation/fraction of functional compounds in natural products because it has various advantages of low temperature and non-toxic solvent residue. A co-solvent can be added to increase polarity and density of CO2 solvent, leading to an increase in extraction yield. Extraction using a co-solvent has been mostly operated via a semi-batch type apparatus. Recently, studies on a continuous extraction have attracted much attention from the viewpoint of throughput and operability by contacting scCO2 with natural compound solution. However, there have been reported a few studies related on the continuous extraction for multi-components systems.
In this work, ethanol and water were used as co-solvents for extraction/separation of perilla components via continuous counter-current scCO2 extraction. Among functional compounds contained in perilla, we focused on perillaldehyde, caryophyllene, squalene and rosmarinic acid, and target compounds in separation of perilla-extract are set to chlorophyll and rosmarinic acid. The former is undesirable compound in final products for its unique color, the latter is valuable compound with anti-dementia effect[1]. Figure 1 shows the results of counter-current extraction at 353 K and 14.0 - 20.0 MPa. The results showed that chlorophyll was more extracted in vapor phase as pressure increased, but rosmarinic acid was not found in vapor phase in this pressure range. Thus, chlorophyll could be separated from rosmarinic acid at high pressure condition.
[1] K. Ono et al., J. Biol. Chem., 287, 14631 (2012)

Wastewater including organic solvent is discharged from the cleaning and drying process in semiconductor manufacturing. It is generally treated by the activated sludge method. However, this method has problems such as a long treatment time, the necessity of pretreatment and aftertreatment, the generation of excess sludge. Therefore, the wastewater treatment technique with a short time and less environmental load is expected. In this study, we focused on catalytic oxidation in superheated steam.
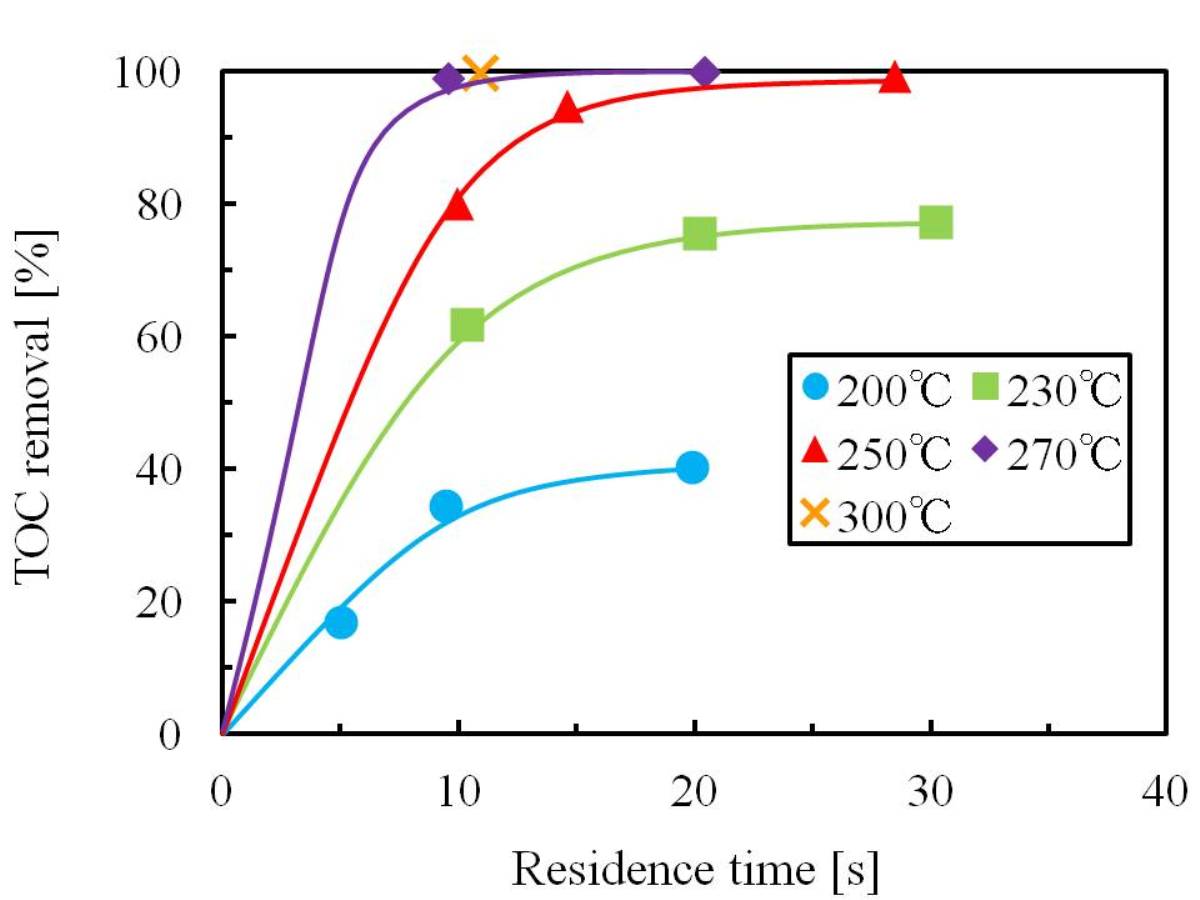
The purpose of this research is the development of wastewater treatment technique by catalytic oxidation in superheated steam. The target substance was isopropyl alcohol (IPA). The experiment was carried out using a plug-flow reactor filled with MnO2 catalyst. The optimum decomposition condition was determined and the kinetics of catalytic oxidation reaction was analyzed. The experimental results were expressed as total organic carbon (TOC) removal.
The dependences of TOC removal on the reaction temperature and residence time were investigated. TOC removal increased with the reaction temperature and residence time. It achieved 99.8 % at 300 °C, 0.9MPa, 11 s and 1.2 of oxidant ratio. On the other hand, TOC removal was constant with long residence time at the low reaction temperatures of 200 °C and 230 °C. It was because acetone and acetic acid were produced by oxidation of IPA, and they were detected as TOC.
The reaction kinetics was examined using the pseudo-first order reaction model, and TOC removal correlated well using this model. Based on the results of this kinetics, the activation energy (Ea) and the frequency factor (A) for TOC decomposition were determined from the Arrhenius equation. As a result, Ea was 56.1 kJ/mol and A was 6.17×104 s-1.
From there results, the catalytic oxidation in superheated steam is useful for wastewater treatment.
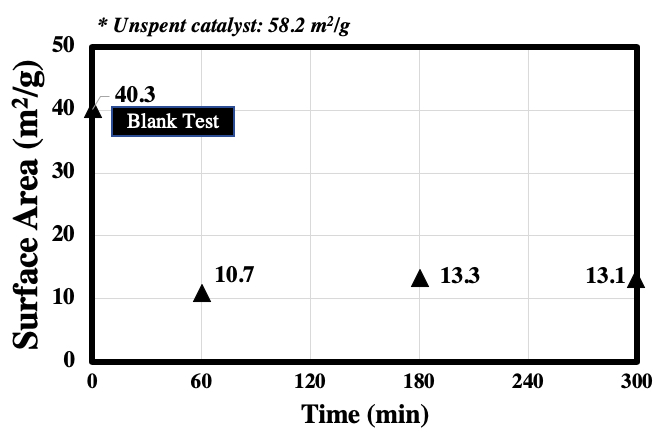
Supercritical water gasification (SCWG) process transforms biomass with high water content into H2-rich gaseous products by utilizing the unique properties of supercritical water at T ≥ 374 °C and P ≥ 22.1 MPa. From the viewpoint of food sustainability, utilizing non-food biomass such as waste cooking oil (WCO) is desirable. So far, its known treatment method is conversion to biodiesel through transesterification and hydrodeoxygenation. However, these processes suffer from low product conversion due to WCO's high water and free fatty acid content. Therefore, SCWG is an attractive option to achieve WCO treatment and bioenergy conversion at the same time. Due to SCWG's high operating requirement, a suitable hydrothermal catalyst is often required. However, the efficiency of this process is currently limited by catalyst deactivation. This study aimed to determine the mechanism of catalyst deactivation in SCWG. Vegetable oil was used as a representative compound for WCO. Experiments were conducted by gasifying vegetable oil using nickel catalysts in lab-scale flow type reactor made of stainless steel tubing. Experimental conditions employed were fixed at 400 °C and 25 MPa at variable feed concentration (2 – 5 wt%) and reaction time (60 – 300 min). The performance of the process was evaluated according to gas yields and efficiencies. Catalyst characterization was carried out before and after the reaction using N2 gas sorption analysis. To probe the catalyst surface, XRD and SEM-EDX analysis were conducted. The results of the study showed that catalyst performance can be observed with the system's ability to convert the biomass into desired gaseous products. Carbon deposition and loss of active catalytic surface area (Figure 1) due to particle sintering were considered as the main causes of nickel catalyst deactivation in the SCWG of vegetable oil.
Food allergy is caused by a specific amino acid sequence of allergen protein. In order to reduce allergenicity of allergen protein, we must destroy specific amino acid sequence by hydrolysis or thermal treatment. Therefore, there are many researches that focus on the amino acid sequence of the protein. On the other hand, it is reported that the molecular weight of the protein determines allergenicity. This is based on a fact that most allergen proteins have a certain range of molecular weight, but there is no scientific basis.
In this study, we focused on the molecular weight of the allergen protein. If the molecular weight of the protein determines the allergenicity, the evaluation of the allergenicity for various proteins become possible because it does not depend on the amino acid sequence of protein.
In Japan, 70% of food allergy are caused by chicken eggs, milk and wheat. Therefore, we selected albumin (chicken egg), casein (milk) and gluten (wheat) as model of allergen proteins. The high-temperature and high-pressure water treatment was applied for the hydrolysis of these protein.
We used a batch reactor for high-temperature and high-pressure water treatment (150 ~ 225 °C and 15 ~ 180 min). The molecular weight was analyzed by size-exclusion chromatography (SEC). The allergenicity was evaluated by Enzyme-Linked ImmunoSorbent Assay (ELISA).
We investigated the relationship between the molecular weight of the hydrolyzed allergen protein and allergenicity. For all the proteins, the reduction of the allergenicity was confirmed with decreasing the molecular weight by high-temperature and high-pressure water treatment. The molecular weight of the hydrolysis product from casein is higher than that from gluten; however, the allergenicity of casein is lower than that of gluten. This is probably due to the difference in the amino acids sequence of the protein.
Extraction of value-added products from biomass such as agricultural residues and food wastes has attracted the attention of persons and organizations interested in establishing a sustainable society. In this study, rice bran and soybean were used for the production of value-added products via supercritical carbon dioxide (scCO2) extraction. The scCO2 extraction was performed at 20 MPa and 40 °C. In the case of rice bran, 15 weight % of dry rice bran was extracted via the scCO2 extraction; 30 weight % of dry rice bran was extracted via the extraction using organic solvent (mixture of toluene and ethanol).
Carbon nanodots (CDs) which are photoluminescent carbon-based nanomaterials doped with nitrogen have attracted much attention to replace inorganic quantum dots because of their excellent properties, such as low cost and low toxicity. For further applications as photoluminescent materials, full color tuning of CDs is required. In this work, CDs were synthesized under various solvothermal conditions with a batch reactor, and relevant parameters for determining the emission wavelength of CDs are discussed.
Phenylenediamines (o-,m- and p-PDAs) was used as the reactant. PDA was dissolved in ethanol at a concentration of 7.3×10-2 M, and sealed in a stainless-steel tube (~10 mL). The high-pressure vessel was placed in an electric furnace, and heated at 150–180 °C for 1–5 h. The crude solution of the product (oCDs, mCDs and pCDs) was diluted by 100, and the emission and excitation spectra and the absorption spectra were measured by a fluorescence spectrophotometer (F-2700, Hitachi). The particle size of CDs was determined from the TEM images. The FT-IR spectra of the CDs were obtained by a spectrometer (FT/IR-4200, Jasco).
The °CDs show green emission at ~540 nm for 350–500 nm excitation, and the pCDs show orange emission at ~580 nm for 370–550 nm excitation, irrespective of the reaction conditions. The mCDs show blue emission at ~500 nm and green emission at ~540 nm depending on the reaction degree. Whereas the particle diameter of these CDs was constant at 2.0–2.5 nm, it was suggested that the chemical structures affect the emission color; suppression of oxidation is related to emission at longer wavelength. The relationship between the optical properties and chemical structure of CDs is discussed in detail at the presentation.
A water-in-supercritical CO2 microemulsion, that is a thermodynamically-stable system of reverse micelles encapsulating aqueous nano-droplets in supercritical CO2, can be prepared by CO2-philic surfactants (e.g. fluorinated surfactants). Due to the solvent properties of water and supercritical CO2, it can be an environmentally friendly and universal solvent able to solubilize both nonpolar and polar substances. However, there are some issues caused by surfactant headgroups in applications of W/C MEs, for example, difficulty to remove surfactants from products, extracts and washings due to a strong surfactant headgroup interactions to those surfaces and compounds.
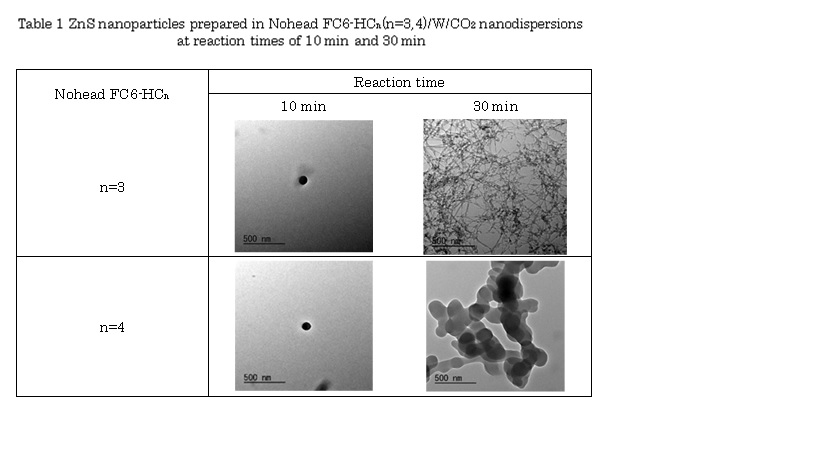
Recently, water-in-supercritical CO2 nanodispersions were successfully prepared by the headgroup-free fluorocarbon-hydrocarbon amphiphile Nohead FC6-HC4 (C6F13-Ph-COC5H11, Ph = phenylene group). The nanostructures of the dispersions were characterized by the high-pressure small-angle neutron scattering (SANS) technique. As the water clusters were able to dissolve water-soluble substances like an ionic dye and salts, it will be a universal green solvent without the issues caused by headgroups. Aiming to develop an effective, efficient and green nanoparticle production technology, this study tested synthesis of inorganic nanoparticles synthesis in water/CO2 nanodispersions with Nohead FC6-HCn (C6F13-Ph-COCH2-CnH2n+1, where n = 3 or 4). Table 1 shows are nanoparticles synthesized in the nanodispersions with 50 mM Nohead FC6-HCn at 200 bar and 45 °C. For ZnS nanoparticles synthesis, the nanoparticles were obtained with reaction times longer than 10 min. Those were spherical with diameters of ~ 100 nm at reaction time of 10 min for both amphiphiles. Interestingly, it became ZnS nanofibers with diameters of ~10 nm for Nohead FC6-HC3 at the reaction time of 30 min, and generated a network structure. Then water/CO2 nanodispersions were found to play a role of nanoreactor.
Nowadays, newly discovered drug candidates which are composed of high-molecular weight compounds have very low solubility in water, resulting in poor or erratic bioavailability. The bioavailability can be improved by reducing the particle sizes of drugs. Supercritical antisolvent recrystallization (SAS) using CO2 has been paid much attention as an effective and environmentally friendly particle design method. SAS process produces particles from a liquid solution by the addition of supercritical CO2 that is partially or totally miscible with the liquid solution and that is a non- or anti-solvent for the solutes. However, conventional SAS processes have problems such as a difficulty of production of small and uniform-sized particles because supersaturation heterogeneity of the solutes in a large volume crystallizer leads to a heterogeneity of nucleation rates and crystal growth rates, resulting in the production of large and ununiform-sized particles. Therefore, we proposed a new SAS using a micro device as a crystallizer (SAS-MD) to solve the problems.
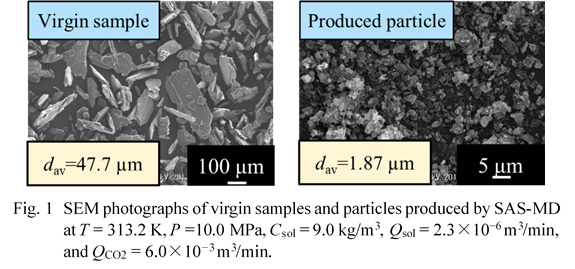
In this work, we aim to elucidate the effects of operation parameters including crystallizer temperatures (313.2–323.2 K), crystallizer pressures (10–19 MPa), liquid solution concentrations (4.0–14.3 kg/m3), liquid solution flow rates (2.3×10–6–2×10–5 m3/min) and CO2 flow rates (3.0×10–3–6.0×10–3 m3/min) on the production of microparticles of sulfathiazole by SAS-MD using a Y-type micro device (internal volume: 1.75×10–10 m3). Scanning Electron Microscope (SEM) images of virgin samples and those produced by SAS-MD are shown in Fig. 1. The particles produced were plate-like or spherical particles with mean sizes of around 2 μm which were nearly 1/22 of a mean size of the unprocessed plate-like samples. SAS-MD caused a polymorph transition from a mixture of form II and form IV to form I. Figure 1 shows the mean sizes of particles by SAS-MD were not mostly dependent on crystallizer temperatures.
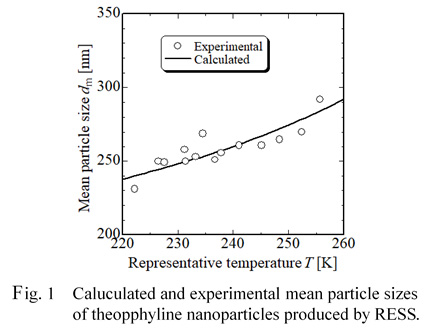
In the pharmaceutical industry, there are great interests in preparation of nanoparticles because particle sizes of drugs are of importance for an enhancement of bioavailability and the application to drug delivery systems (DDS). New process using supercritical fluids have been expected as an effective and environmentally friendly nanoparticle design process. The most well-known micronization technique based on supercritical techniques is the rapid expansion of supercritical solutions (RESS). In the RESS technique, a solute is dissolved in a supercritical fluid, and the solution is then expanded rapidly through an expansion nozzle. Because of the large rapid drop in both pressure and temperature, the solute dissolved becomes insoluble in a low-pressure gas and thus a large degree of supersaturation is created in the spray jet. Rapid nucleation and growth of crystalline particles occur, followed by production of micro- or nano-sized particles due to a very high supersaturation and a very small growth time. We have successfully produced nanoparticles with mean sizes ranging 200 to 700 nm of several drugs including ibuprofen, theophylline, caffeine and phenacetin, and have elucidated effects of several operation parameters in the RESS on the production of the nanoparticles. For practical application of this technique, it is necessary to establish particle design technique.
In this work, we report on a new and simple model to predict mean particle sizes of nanoparticles of several drugs produced by RESS. This model is based on both the mass balance in the RESS process and the classical nucleation theory and has two parameters including supersaturation and a representative temperature which is an average temperature between the temperature at inlet and that at outlet of the crystallization chamber. Figure 1 shows experimental and calculated results of mean particle sizes of theophylline nanoparticles produced by RESS and shows the model give good predicted results.