
Two-dimensional (2D) nanosheet membranes are expected to function as size-selective molecular separation membranes, based on their unique atomic thickness with micrometer lateral dimensions. Stacked nanosheet membranes are formed by assembling single molecular sheets into thin membranes, and contain 2D nanochannels between the stacked sheets that allow water or solvents to pass through whilst rejecting unwanted solutes [1]. Therefore, they represent promising materials for potentially high-functional membranes for liquid separation such as water treatment and organic solvent filtration. The structural stability of nanosheet membranes during filtration is also a critical issue for their application.
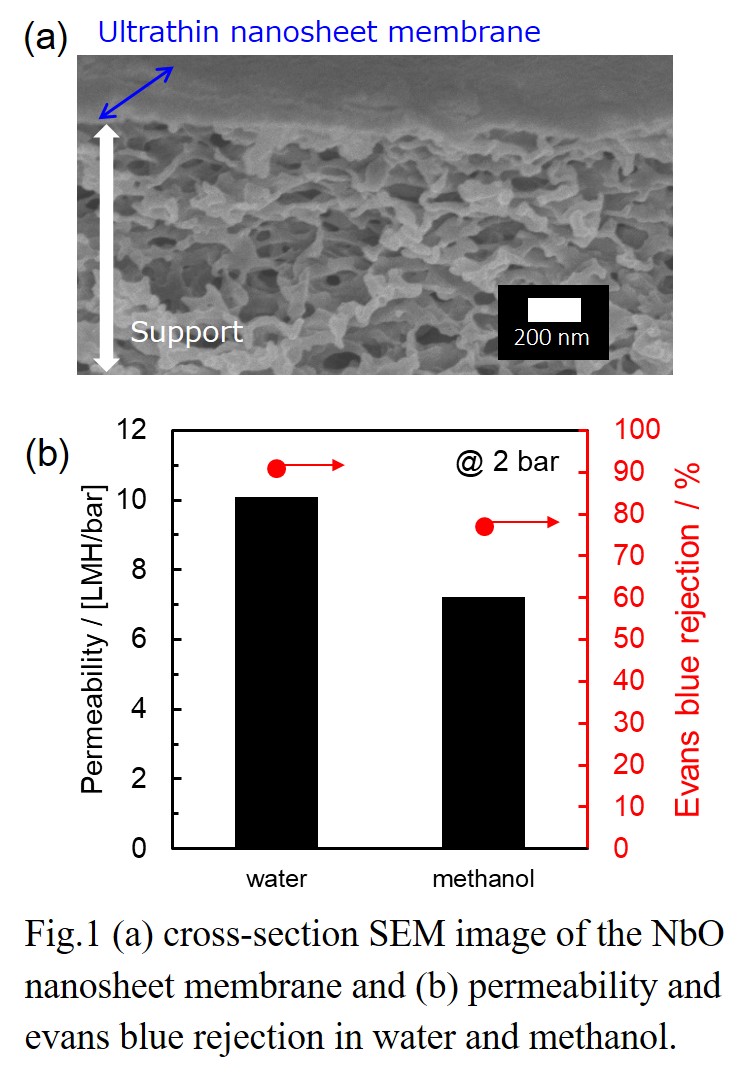
In this study, we fabricated nanosheet membranes using single 2D nanosheets of niobium oxide (NbO) on a porous cellulose nitrate or anodic alumina support by a simple vacuum filtration. The thickness of NbO membranes were controlled by adjusting the volume of nanosheet colloidal solution used during the vacuum filtration. The stacked NbO nanosheet membranes had a dense structure and were highly stable during separation tests, because of the chemical cross-linking between nanosheets. The method allowed the formation of nanochannels in the NbO membranes. The NbO membranes showed high rejection performances against anionic dyes and salts in water [2,3]. Furthermore, membrane structure and separation performances in alcohols were also investigated. There was little difference in the interlayer spacing of the stacked structure between water and alcohols, suggesting the stable layered structure in alcohol solvents. The NbO membranes demonstrated relatively high rejection performances such as 85% rejection for Evans blue (EB, Mw: 960.8) in methanol. The channel structure and separation mechanism for NbO membranes in water and alcohols will be discussed.
[1] G. Liu, et al., Angew. Chem. Int. Ed.55, 2–16 (2016), [2] K. Nakagawa et al., Chem. Commun. 53, 7929-7932 (2017), [3] K. Nakagawa et al., Sep. Purif. Technol, 219, 222-229 (2019)
High purity propylene is an octane-enhancing chemical and also feedstock to industrially important chemicals. Purification of propylene from propane mixture is technologically and financially challenging because of their close boiling points. ZIF-8 membrane has the potential to separate propylene from propane effectively due to the gate opening effects, although the theoretical ZIF-8 aperture size (0.34 nm) is smaller than the kinetic diameters of propylene (0.4 nm) and propane (0.43 nm). In this work, defect free ZIF-8 membranes were successfully developed from the secondary growth seeding technique with sodium formate as deprotonating agent that facilitated continuous, well-intergrown ZIF-8 membrane on α-Al2O3 support. The defects formed by the crack formation in the membrane was steadily and effectively removed by the uniquely discovered self-healing property that the sodium formate extended. The ZIF-8 membranes demonstrated their excellent molecular sieve separation capability for equal molar propylene/propane mixture with the highest separation factor of 115 and average propylene permeance of 50.40×10-10 mol/m2 s Pa.
In this talk, I will report the development of novel omniphobic membranes and operation modes for various process applications with membrane contactors (MCs). An omniphobic membrane was fabricated for membrane distillation (MD) by effectively depositing ZnO nanoparticles on a hydrophilic glass fiber (GF) membrane to create hierarchical re-entrant structures, followed by surface fluorination and the addition of a polymer coating to lower the surface energy of the membrane. The omniphobic membranes possessed a particulate membrane morphology and an extremely high fluorine concentration on the surface. The omniphobicity of the fabricated membrane was indicated by the contact angles for water and ethanol, which were as high as 152.8±1.1 ° and 110.3±1.9 °, respectively. In particular, it will focus on the development of tuned hydrophobic, hydrophilic and asymmetric wettability membranes. Membrane contactors are membrane systems that can find application in different fields of industrial interest, covering, for example, gas-liquid operations, liquid-liquid extractions and vapor-liquid distillation. In past decade, the use of membrane absorption to capture carbon (membrane absorption, MA), to prepare emulsions (membrane emulsification, ME), to recover resources from sea (membrane crystallization, MCr) to carry out distillation processes for water scarcity issue (membrane distillation, MD) has been subject of many research activities worldwide. Focus of this talk will be placed on the applications of the developed novel aerogel membranes for MA and MD processes to resolve the carbon abundance and water scarcity issues, respectively, for achieving an ultimate goal toward a sustainable planet.
Silica membranes have been developing as hydrogen permselective membranes. A counter diffusion chemical vapor deposition (CVD) method is the preparation method for the silica membranes. One of the problems for the application of hydrogen permselective membranes is hydrothermal stability. In this study, effects of organic groups in the silica structure on hydrothermal stability was investigated.
γ-alumina (マ・0 mm, L30 mm, Noritake Co..) was used for a porous substrate. γ-alumina layer or silica sol-gel layer was coated on the porous substrate. Aluminum sol 5S (Kawaken Fine Chem. Co) with polyvinylalcohol (PVA) was used for the coating of the γ-alumina layer. The parent sol of Tetraethoxysilane (TEOS):EtOH: H2O:HNO3=1:0.1:4:5 was employed for the silica coating. The coated substrates were calcined at 600 °C. CVD was carried out by using HTMOS (Hexyltrimethoxysilane) or TMOS (Tetramethoxysilane) as a silica source at 450 or 600 °C for 15 min. Single gas permeation tests were carried out by using H2 and SF6 at 270 °C.
The hydrothermal stability under steam was examined for the TMOS derived membrane deposited at 550 °C and for the HTMOS derived membrane deposited at 450 °C. The time courses of H2 permeance through the both membranes were shown the figure. The H2 permeance through the TMOS derived membrane decreased gradually, and the permeance was reduced by 37% under the steam treatment for 12h. On the other hand, the reduction rate of the H2 permeance through the HTMOS derived membrane on the silica coated substrates after 23h of the steam treatment was only 10%. In order to confirm the effects of coating of the substrates, the results through the membrane deposited on the coating of the γ-alumina layer were also shown as open plots in the same figure. The H2 permeance looks the similar indicating that there were little effects on the HTMOS derived membranes.
Membrane separation technology has been paid attention for one of the energy saving technologies. A silica membrane is categorized as one of the typical inorganic membranes, and the silica membranes show high hydrogen permselective performance comparing with the other inorganic membranes, such as zeolite and carbon membranes. On the other hand, it is generally known that permselective performance of silica membranes are affected strongly under hydrothermal conditions.
In this study, influence of steam on permeation performance of the DMDPS-derived silica membrane was evaluated to develop the silica membranes having relatively high hydrothermal durability. The silica membrane was prepared by counter-diffusion chemical vapor deposition method on a porous alumina support purchased from Nikkato co., Japan. After membrane preparation, permeation performance was measured using single component H2, N2 and SF6, respectively. Heat-treatment of the membrane was conducted at 573 K for 50 h. During heat-treatment, H2, N2 and SF6 permeances showed approximately constant value. To evaluate effect of heat-treatment, durability test was conducted under 0.1 ~ 1.0 mol% steam co-existed conditions at 573 K by using heat-treated and non-treated silica membranes. Comparing influence of steam on permeation performances of these membranes, permeance decreasing ratio of heat-treated membrane showed lower value than that of non-treated membrane in any steam concentrations. In addition, in-situ diffuse reflectance FT-IR spectra of heat-treated and non-treated silica powder was obtained. The heat-treated silica had less amount of adsorbed water molecules than the non-treated silica because the silanol group was decreased owing to heat treatment. From these results, it is considered that hydrothermal durability of the silica membrane is determined by the silanol group density.