
Food flavor is an important factor in determining the appetite and taste of food. The production of the emulsified flavor powders and the controlled flavor release from the powders have been extensively investigated. Spray drying is one of the most widely used processes for producing powders containing emulsified flavor in food industry. Flavor encapsulation and the release rate are significantly affected by wall material and emulsifier. Wall materials are one of the most important factors which may influence the barrier properties against the flavor diffusion in spray-dried powder. In this study, release rates of d-limonene from spray-dried powders were investigated using various wall materials, such as maltodextrin (MD) (dextrose equivalent (DE) = 25 and 19), lactose (Lac), and sucrose (Suc). d-Limonnen and hydrolyzed casein protein (Emulup) were a core material and an emulsifier, respectively. A pilot-scale spray dryer (L-8 type, Ohkawara Kakohki Co., Ltd.) was used to produce spray-dried powders. Two techniques were employed for measuring the release rate of d-limonene; a hand-build DVS system assembled with GC-FID detector under a ramping humidity condition for short term releasing experiment, and the conventional method by which the powders were placed in a desiccator keeping at constant temperature and relative humidity (RH) for long term releasing experiment. For the conventional method, the release rate constant of d-limonene were estimated by applying the Avrami equation. Logarithm of the release rate constants were proportionally increasing to RH for Lac as well as for MD. For the ramping humidity experiment, a linear correlation was found between the humidity inducing release of d-limonene from the powders and the glass transition temperature of wall materials. The release rates were significantly dependent on the wall material.
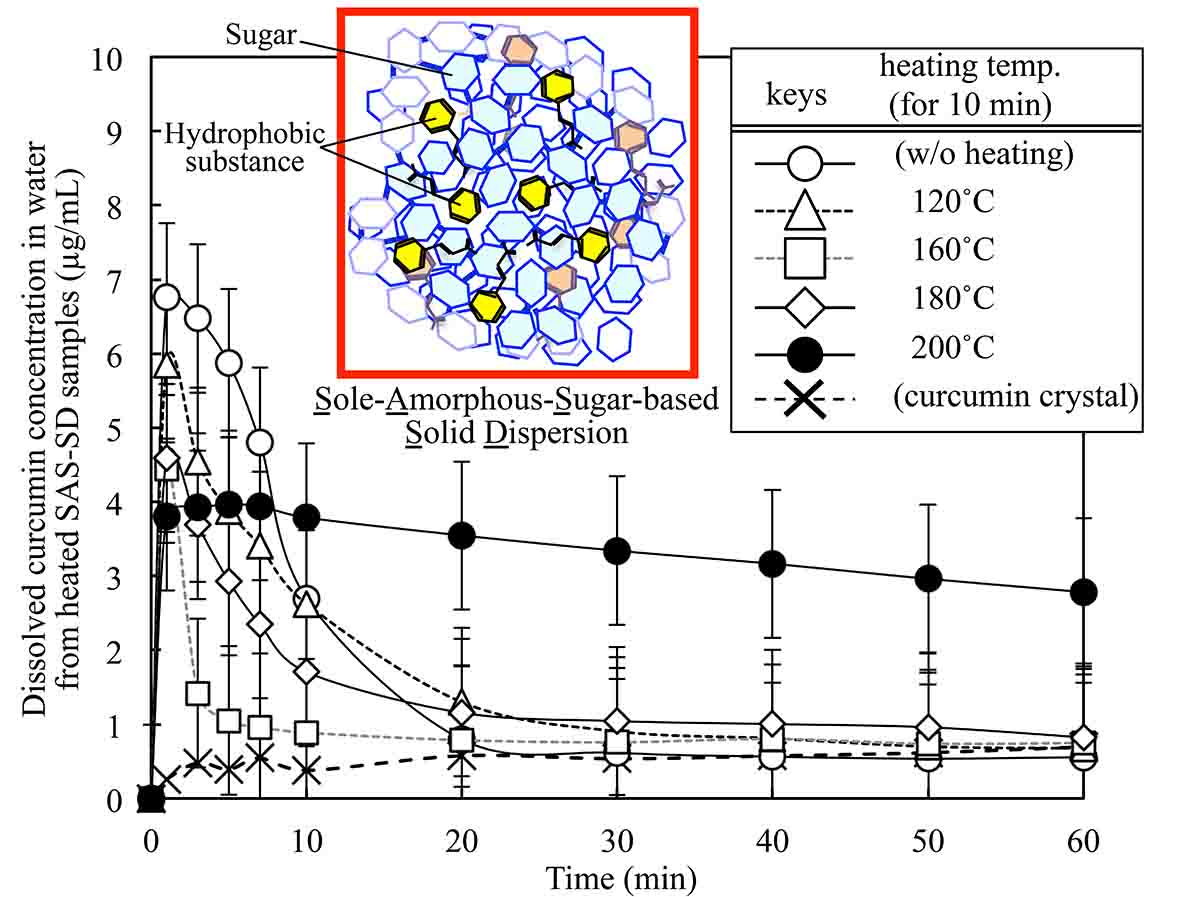
To date, numerous attempts have been made to improve the solubility of hydrophobic ingredients in physiological fluids. One such method for improving the water solubility of a hydrophobic substance is the use of an “amorphous solid dispersion.” We recently developed a new amorphous solid dispersion technique that does not involve the use of a surface active agent (Satoh et al., Food Chem. 2016, 197: 1136–1142). In this method, (i) sugar is amorphized and (ii) added to an organic solvent containing a hydrophobic substance, followed by homogenization. (iii) The homogenized solution is then dried to a solid. This sole amorphous sugar-based solid dispersion (SAS-SD) technique was applied to curcumin, known as food additive, anti-inflammatory, antitumor, anti-amyloid agents. The SAS-SD of curcumin was prepared by vacuum foam drying from methanol, using disaccharides as drug carrier-forming component. The analyses of microscopic morphology and thermal characteristics of the SAS-SD indicated the plasticizing effect of curcumin on the carrier sugar matrix. Aqueous dissolution profiles of curcumin from the SAS-SD were compared among different sugar types and sugar/curcumin compositions. All the SAS-SD samples exhibited “spring & parachute” type dissolution profiles of dissolved curcumin concentration. Markedly increased dissolution, represented by the area under the time course curve of dissolved curcumin (AUC60min), was observed when α-maltose was added in the composition range equal to and above 100 g-sugar/g-curcumin. Furthermore, heating the SAS-SD at close to and above the melting points of curcumin significantly prolonged the curcumin dissolution in water, resulting markedly increased AUC60min value.
Reported phosphate binders for the treatment of hyperphosphatemia have high pill burden, cost and associated disadvantages like deposition in bones and adverse health effects. In present report, a synbiotic preparation was formulated using Lactobacillus casei JCM 1134 and deacetylated Aloe Vera (DAV) and evaluated its phosphate accumulation efficiency with the commercials. Aloe Vera powder was boiled in water bath at pH 8.0, 65 °C for 12 min. The centrifuges pellet was considered as DAV. Under optimized conditions, L. casei and DAV were mixed with additives and kept for incubation of 3h at 37 °C. Later the mixture was spray dried with supernant of DAV preparation and a synbiotic spray dried powder (SD) was obtained. Milk and coca-cola were taken as standard phosphate rich food. CaCO3, LaCO3, Al(OH) 3 and SD were studied as phosphate binders. In-vitro broth study was conducted and supernant was assayed for remaining phosphate using ascorbic acid method. Spray drying increased phosphate accumulation capability of L. casei in in-vitro broth. SD was observed as the second best after CaCO3 (accumulated phosphate 95% and 93% w/v from milk and coca cola respectively) and accumulated phosphate around 88% and 87% w/v from milk and coca cola respectively. However, LaCO3 and accumulated phosphate ~ 67% and 37% w/v while Al(OH)3 accumulated phosphate 83% and 73% w/v from milk and coca cola respectively. For safety, efficiency and cost, SD could be the best answer for preventing hyperphosphatemia. As CaCO3 is the best among tested phosphate binders, but it conferred hypercalcemia and gastrointestinal side effects etc. However, SD is composed of probiotic and Aloe vera which have GRAS status, and therefore, it is safe, affordable and comparatively effective as CaCO3.
There has been growing interest in gaining insights into the design of novel foods whose digestibility is controllable based on life stages and health conditions. Physical digestion is especially important in the case of solid foods, since their size reduction caused by breaking down can promote chemical and enzymatic digestion and release of nutrients simultaneously. Gastric digestion is the major digestion process in human gastrointestinal tract. The development of simple in vitro gastric digestion devices that enable quantitative analysis of the digestion dynamics of solid foods has been necessary. The authors and co-workers developed a human gastric digestion simulator (GDS) that simplifies the antrum geometry, is capable of simulated gastric peristalsis, and which enables direct observation of the disintegration of food particles in the gastric contents. This presentation provides a brief overview of our findings regarding the digestion dynamics of solid foods by GDS analysis. First, the concept and development of the GDS is introduced. The disintegration characteristics of solid foods using the GDS are described next, providing insights into the digestion processes influenced by gastric peristalsis. The GDS is expected to offer potential as a tool for designing novel nutraceutical and functional foods for which digestibility is well controlled.
A freeze-dryer with a radiative heating device was constructed and the inner vapor transfer property was regulated by separating the drying chamber and the condenser with an orifice. It was confirmed that the inner vapor transfer property substantially affected the acceptable operation range for avoiding product collapse, and it was revealed that this coefficient was key to quantify the drying performance of a freeze-dryer. Model foods that contained flavor components (i.e. saturated linear alcohols and ethyl esters) were freeze-dried under various operative conditions and the retention of those components were evaluated. It found that the inner vapor transfer property significantly affected the retention of the flavor components in relation to the dried cake properties. A mathematical model was used to obtain the contour diagrams of the selected parameters (e.g., product temperature and total drying period) during drying as a function of the heating condition and chamber pressure. The inner vapor transfer coefficients of the freeze-dryer were used to draw the operation lines on the diagrams. An attempt was made to depict the retention of the flavor components in combination with the contour diagrams and operation lines. It could be a rational method to predict end-product quality of a dried food based on mechanistic mathematical model.
Enzymatic hydrolysis process of polysaccharide is frequently used in food industries. Generally, small amounts of enzyme is added into the substrate solution or suspension. Efficient mixing in such system is quite difficult. Moreover, if a strong shear force is applied to enzyme, the three-dimensional structure of enzyme breaks down. This leads the deactivation of enzyme. Therefore, a reactor having the efficient and gentle mixing characteristic have been required.
In this study, Taylor-Couette flow reactor (TCFR) consists of two concentric cylinders with the inner one rotating was applied to hydrolysis process of polysaccharide. Above a critical Reynolds number (Recr) in the circumferential direction, there appear pairs of counter-rotating toroidal vortices (so called Taylor vortex flow) spaced regularly along the cylinder axis. The toroidal motion of Taylor vortex flow enhances not only heat and mass transfer but also mixing without the locally strong shear force. Furthermore, all the fluid elements leaving the annulus have the same residence time when the relatively small axial flow is added. In this study, in order to optimize enzymatic hydrolysis process of polysaccharide using TCFR, the effect of the rotational speed of inner cylinder and the axial flow rate on the yield of sugars was investigated.
The yield of sugars increased with the increase in circumferential Re, but it decreased in highly Re region where a wavy motion occurs. In wavy Taylor vortex flow region, the axial dispersion is promoted while the intra-mixing in Taylor cell is enhanced. This axial dispersion would induce the decrease in the yield of sugars. Thus, it can be concluded that TCFR should be operated in laminar Taylor vortex flow region in enzymatic hydrolysis process of polysaccharide.