
Alginate and chitosan have favorable properties as biomaterials such as nontoxicity, biodegradability, biocompatibility and antibiotic property. Alginate and chitosan ultrafine fibers generated through electrospinning have drawn widespread attention at tissue engineering scaffolds, drug release carriers, wound dressings and so forth. While both alginate and chitosan are water-soluble, the electrospun mats have to be insolubilized before the usage in these applications. Herein, we proposed the method of preparing alginate and chitosan water-insoluble nanofibers through electrospinning and horseradish peroxidase (HRP)-mediated crosslinking of phenolic hydroxyl (Ph) moieties under stimulating of vaporized H2O2. In details, alginate and chitosan were firstly modified with Ph moieties, namely alginate-Ph and chitosan-Ph. Alginate-Ph blending (alginate-Ph 2% w/v, polyethylene oxide (PEO) 9% w/v, Pluronic F-127 5% w/v, HRP 160 U/ml ) and chitosan-Ph blending (chitosan-Ph 1% w/v, PEO 10% w/v, Pluronic F-127 10% w/v, HRP 160 U/ml ), were electrospun separately to fabricate nanofibers under conditions of voltage 11~15kv, needle to collector distance 8~15cm and flow rate 0.1 ml/h. The resultant fibers were then exposed to the air containing 3 ppm H2O2. The diameters of alginate-Ph fibers before and after exposure to the air containing H2O2 were 171 nm and 200 nm, respectively. Similar results were observed in the case of using chitosan-Ph. Successful crosslinking was confirmed for both alginate-Ph and chitosan-Ph fibers from the result of keeping fibrous structure after 48 h of soaking in PBS. In contrast, the fibers non-exposed to the air containing H2O2 dissolved immediately after soaking in PBS. These results clearly indicated the possibility of obtaining insolubilized nanofibers through electrospinning and HRP catalyzed reaction in the air containing H2O2.

Endoscopic submucosal dissection (ESD) for the treatment of esophageal mucosal lesions is associated with high rates of postoperative stenosis, resulting in significantly decreased quality of life. Several therapies are available for the prevention and treatment of esophageal stenosis after ESD, including prevention via pharmacological treatment, endoscopic esophageal dilatation, stent placement, and autologous cell transplantation. However, no single method has been currently recognized as effective in clinical practices. Therefore, simple yet effective preventive methods are crucial to reduce the risk of postoperative esophageal stenosis.
Considering the operability under the endoscope, a spray material that can be administered through a 1.5 m-long and 4 mm-diameter endoscopic instrument channel is expected to be promising. Therefore, in this research, we developed a novel in situ cross-linkable powder for the prevention of esophageal stenosis after ESD. The powder was composed of alginate and gelatin; alginate can be rapidly cross-linked by calcium ions, whereas gelatin can be cross-linked with collagen on the submucosal exfoliated surface by transglutaminase. Therefore, by spraying them with their cross-linker solutions, hydrogel can be formed immediately with a firm adhesion to the wound surface.
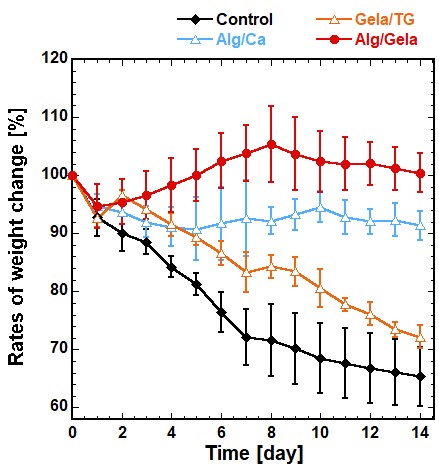
Because there was no appropriate small animal model for evaluating esophageal stenosis-preventing material, we developed a brand-new stenosis model in rats by combining mechanical and trypsin treatment to rat esophagus. The rat model successfully caused stenosis, which was close to ESD-induced stenosis in clinical. The result of material administration to this model is shown in the figure below. The body-weight loss caused by stenosis was clearly reduced by the application of alginate/gelatin-mixed powder compared with untreated control group. The esophageal patency rate after 1 week and 2 weeks after surgery was also significantly improved compared with the control group. From these results, it is expected that this sprayable material is useful for preventing esophageal stricture after ESD.
The prospect of regenerating organs and tissues would bring new possibilities of improving current treatments or find solutions for untreatable situations, thus having an enormous impact in the quality of life of patients. Tissue Engineering has been integrating principles of engineering, chemistry, materials science, biology and health sciences in order to develop regenerative-based therapeutic strategies combining stem cells and biomaterials. From the different sources of biomaterials, polysaccharides have been proposed to produce matrices able to interact favourably with cells. Due to their hydrophilic nature and richness in chemically active groups, such polymers can be used to produce a variety of structures fabricated using aqueous-based or other environmental-favourable procedures. Examples are shown in the modification of polysaccharides from marine origin and processing of devices into different sizes and shapes (e.g. hydrogels, multilayered coatings and membranes, particles) with structural and functional characteristics suitable to be used in tissue engineering and regenerative medicine applications. In particular, distinct strategies involving bioinspired approaches and nano/micro-technologies will be shown to engineer hybrid soft systems containing cells, in the form of microparticles, microgels or thin films, that could give rise to biomedical devices using bottom-up strategies.
Human skeletal muscle myoblast (HSMM) sheet transplantation is promising therapy for severe heart failure. The myoblast sheet has proved to secrete multiple cytokines such as vascular endothelial growth factor (VEGF), hepatocyte growth factor (HGF) and fibroblast growth factor-2 (FGF-2), which associate with cardiac functional improvement. The purpose of this study is to explore the regulation of cytokine productions and the impact of human skeletal muscle fibroblasts (HSMFs) on the regulation in HSMM sheet. Our current results demonstrated that cell density/myo-myo contact regulate the VEGF production in HSMMs. The VEGF productivity was high at low density in HSMM culture and vice versa while the productivity was low and unchanged by cell density in HSMFs. By varying ratio of HSMFs, it was shown that the VEGF productivity was increased by 2-fold (maximum level) by addition of small population of HSMFs (at 15%) in HSMM culture but the productivity was more reduced at higher ratios of HSMFs. For HGF, the productivity was HSMF ratio dependency and HGF was undetectable in HSMM culture alone suggesting that HGF was mainly secreted from HSMFs. Furthermore, FGF-2 was undetectable in all conditions. By using a cell tracking system and time-lapse observation, we found that HSMFs showed fast-random migration and disrupted myoblast alignment in HSMM culture. This evidence implied that a minor population of HSMFs in HSMM culture might promote VEGF productivity through physical myo-myo contact disruption. Our studies provided the fundamental understanding of VEGF regulation, which is useful to develop complex HSMM sheet or engineered tissue for transplantation, and highlight the role of fibroblasts on VEGF secretion regulation from adjacent tissue in pathological conditions.
Islet allotransplantation is a forthright approach to restore normoglycemia without daily, lifelong insulin injections. Nevertheless, the majority of the islets are lost almost immediately after transplantation due to poor access to nutrients from lack of vascularization which results in need of multiple donors for a single operation. In order to address this issue, highly-porous polymeric scaffold was fabricated as an implantable bioartificial pancreas utilizing modular tissue engineering approach. Pancreas β-cells were immobilized in three-dimensional scaffold and studied for its functionality as bioartificial pancreas in 7-day perfusion culture. During the 1-week perfusion culture, the amount of glucose consumption was observed decreasing and the produced lactate remained relatively low and constant in the cell-laden micro-scaffolds. Referring to the glucose-dependent function, the insulin secretion kept increase during the perfusion culture. Additionally, as visualized by live/dead staining, the immobilized cells remained viable after 7 days of perfusion. The β cells seemed to persist in clusters rather than separated single cells. These results demonstrated the potential of the micro-scaffold to maintain aerobic respiration for cultured cells, which could be resulted from the highly porous structure of the constructs.